Document Type : Original Research Article
Authors
Department of Chemistry, Payame Noor University, Tehran, Iran
Abstract
A new and efficient graphene oxide catalyst was synthesized in this study to prepare trisubstituted imidazoles. Graphene oxide was first modified with 2-chloroethanol, and then treated with N,N,N,N-tetramethylethylenediamine. After that, the obtained catalyst was characterized by XRD, FTIR, FESEM, EDAX, and TGA. The reported catalyst was utilized for the synthesis of trisubstituted imidazoles from benzil, aldehydes, and ammonium acetate. Reaction efficiently proceeded via a one-pot multicomponent route under solvent-free conditions. Catalyst offered some advantages over reported ones such as sustainability, cost-effectiveness, ease of handling, storage, and recovery, in addition to non-metal structure. The reported procedure has a series of benefits, including medium conditions, high yields, no by-products, short reaction times, and easy work-up. Reported catalyst can be used in chemistry, material sciences, and related disciplines for diverse applications.
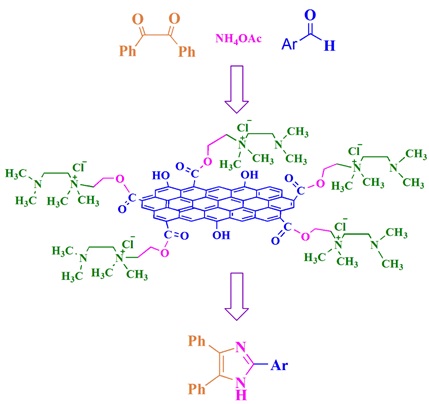
Graphical Abstract
Keywords
- Graphene oxide 2
- Chloroethanol Ionic liquid Tetramethylethylenediamine Trisubstituted imidazole Green chemistry
Main Subjects
Introduction
Catalysts are critical in the sustainable synthesis for several advantages, mainly environmental protection in the chemical industry. Chemistry and sciences focus on sustainability to design modern life without harming the ecosystems [1]. Non-metal and carbon-based catalysts are mainly environmentally benign. Numerous carbon-based catalysts have been designed and used for the synthesis of various chemicals [2], among which graphene and graphene oxide are two necessary carbon materials with excellent properties, including flexibility for designing new and safe catalytic systems [3].
Multicomponent reactions paly an essential role in the design of sustainable reactions with better selectivity and yield and fewer by-products [4-6]. The mentioned reactions are the best alternative for multistep reactions in designing safe organic reactions [7-9]. Another protocol for designing sustainable reactions involves no use of solvents, as toxic solvents are the primary source of harming the environment [10, 11]. Trisubstituted imidazoles are an essential class of heterocycles with a series of properties. A series of biological properties have been reported for them, for instance, antifungal, antitumor, analgesic, and antibacterial [12]. Some derivatives accelerate biosynthesis of interlukin-1, used as glucagon receptors, herbicide, and fungicide [13]. Then can be further used in material science as light-sensitive materials as blue-light emitting materials, sensors, and fluorescent agents [14]. The significant properties of trisubstituted imidazoles have been addressed [15]. The one-pot, three-component synthesis via diketones, ammonium acetate and aldehydes are commonly used in the presence of appropriate catalysts [16]. Numerous catalysts including CoFe2O4@SiO2@(–CH2)3OWO3H NPs [17], graphene oxide-substituted sulfoacetic acid amide [18], NCP@SiO3PrNHPrSiO3TiO2 [19], graphene oxide functionalized diethanolamine sulfate [20], GO-TiO2 nanocomposite [21], sulfated magnetic cellulose nanoparticles [22], trifluoroacetic acid supported starch/graphene oxide nanocomposite [23], zirconium-based metal–organic framework (Zr-MOF) nanocomposites [24], RGO supported Au nanoparticles [25], sulfated polyborate [26], BNPs@SiO2-TPPTSA [27], and magnetic CoFe2O4/Ni-BTC based MOF composite [28] have been explored. These procedures suffer from some several drawbacks such as long reaction times, the use of toxic catalysts and metals, vigorous reaction conditions, low yields, boring purification process, and high amounts of wastes and by-products, necessitating the development of more sustainable procedures.
In this study, graphene oxide-substituted tetramethylethylenediamine ammonium salt was prepared as an ionic catalyst and its activity was examined for the synthesis of 2,4,5-trisubstituted imidazoles under solvent-free conditions with excellent yield.
Experimental
Reagents and chemicals were purchased from Merck chemical company which used as received. The products were characterized by comparing their melting points with previously reported ones. Silica gel SIL G/UV 254 plates were used to study the progress of reactions. JASCO FT-IR spectrometer was employed to record FT-IR spectra using KBr pellets. Melting points were also determined by an electrothermal apparatus. Bruker Avance DRX 400 MHz spectrometer was utilized to afford 1H-NMR and 13C-NMR spectra. Shimadzu model: XRD 6000 apparatus was used to record XRD patterns. The FESEM images and EDAX studies were further obtained using a TESCAN electron microscope. Thermogravimettric analysis (TGA) was conducted on catalyst using a TA apparatus (model Q600) in the range of 30 °C to 500 °C and heating rate of 10 °C.min-1 under argon atmosphere.
Synthesis of graphene oxide substituted 2-chloroethanol (GO@CE)
Graphene oxide was prepared according to the modified Hummers method [29]. Graphene oxide (1.0 g) was suspended in THF (30 mL) and the mixture was sonicated for 60 min. to afford a colloidal solution. Then, 2-chloroethanol (0.7 mL), triethylamine (0.8 mL) and DCC (0.5 g) was added to the solution. The reaction mixture was stirred at room temperature for 30 h followed by adding DMSO (30 mL) and hot filtration. The precipitate was washed with hot ethanol, hot deionized water and acetone several times and dried to afford GO@CE as a black fine powder.
Synthesis of [email protected] catalyst
Dimethylacetamide (30 mL) was added to GO@CE (1.0 g) and sonicated for 60 min. to afford a colloidal solution, and then tetramethylethylenediamine (0.5 mL) was added and the mixture was heated at reflux conditions for 24 h. Finally, the mixture was filtered and the precipitate was washed with dry chloroform several times, dried, and stored in a tight vial.
General procedure for the production of trisubstituted imidazoles using the catalyst
To benzil (1 mmol), aldehyde (1 mmol), ammonium acetate (2 mmol) and catalyst (0.02 g) were added. The mixture was heated in an oil bath for appropriate time and after completion (as indicated by TLC using n-hexane/ethyl acetate (3:1)) ethyl acetate was added and filtered hot. The catalyst was collected by the filter paper and mother liquor was stored at room temperature to crystallize trisubstitued imidazole product. The catalyst was recovered and reused for five runs (Figure 7).
Spectral data of 3k
1H-NMR (400 MHz, DMSO-d6) δ 5.33 (s, 2H), 7.12-7.14 (m, 2H), 7.20-7.49 (m, 10H), 7.74 (t, J = 4 Hz, 2H), 8.00-8.02 (m, 2H), 8.25-8.27 (m, 2H), and 12.51 (s, 1H) ppm. 13C-NMR (100 MHz, DMSO-d6) δ 158.5, 147.5, 146.0, 145.3, 136.8, 132.0, 128.8, 128.8, 128.7, 127.4, 127.3, 124.2, 124.0, 122.4, 115.5, and 68.6.
Results and Discussion
Preparation of catalyst
Graphene oxide was prepared according to the modified Hummers method [29]. It was then functionalized with 2-chloroethanole in the presence of triethylamine and DCC in THF solvent (Scheme 1). The mixture was purified using filtration and washed with deionized water and ethanol, dried and treated with tetramethylethylendiamine in dimethylacetamide to afford catalyst (Scheme 1).
Characterization of catalyst and precursors
Intermediates and final catalyst such as graphene oxide (GO), 2-chloroethanol-functionalized graphene oxide (GO@CE), and [email protected] catalyst were characterized by reliable analytical methods such as FT-IR, XRD, FESEM, EDS, and TGA.

Scheme 1. Production of [email protected]
FTIR spectra
FTIR spectrum of graphene oxide showed stretching vibrations of carboxylic acid hydroxyl groups at ~ 3419 cm-1 and vibrations of carbonyl groups at 1720 cm-1. Moreover, the vibrations of etheric groups (C-O) appeared at 1177 cm-1 [30]. FTIR spectrum of GO@CE is depicted in Figure 1a which shows frequency vibrations of hydroxyl of carboxylic acid groups at 3430 cm-1 [31], and vibrations of hydroxyls at 3328 cm-1 [32]. Furthermore, vibrational frequencies of CH groups were detected at 2851 cm-1 and 2926 cm-1 [33]. The peak appeared at 1727 cm-1 can be also assigned to the carbonyl groups [34], while the bending vibrations of O-H appeared at 1626 cm-1 [35]. The peak at 1575 cm-1 indicates the vibrational frequencies of C=C bonds [36]. Furthermore, vibrational deformation of CH2 was detected at 1439 cm-1 [37], whereas the bending vibrations of C-N are manifested at 1312 cm-1 [38]. Moreover, the band at 1241 cm-1 can be assigned to C-O-C bonds [39], while the frequencies at 1089 cm-1 can be attributed to C-O bonds [40]. Furthermore, the peak at 649 cm-1 can be ascribed to C-Cl bonds [41].
FTIR spectrum of [email protected] catalyst can be found in Figure 1b in which the vibrations of carboxylic acid OH bonds appeared at 3431 cm-1 [31]. Likewise, frequency vibrations of aliphatic CH bonds emerged at 2925 cm-1 and 2856 cm-1 [33]. Vibrations of carbonyl groups emerged at 1713 cm-1 [34], while the vibrations of C-N and C=C bonds appeared at 1629 cm-1 [42]. Moreover, aromatic C=C vibrations appeared at 1573 cm-1 [43], whereas asymmetrical vibrations of CH2 emerged at 1463 cm-1 [44]. Furthermore, the peak at 1112 cm-1 can be assigned to COO of ester, C-N and C-O ether bonds [45]. The FTIR spectra confirmed the structure of GO@CE and [email protected].

Figure 1. FT-IR spectra of a) GO@CE and b) [email protected]
XRD patterns
XRD patterns of GO, GO@CE, and [email protected] are presented in Figure 2. Based on Figure 2a, XRD of graphene oxide showed a sharp peak at 11o and a broad peak, determining reduced graphene oxide between 18° to 22o [46, 47]. Moreover, the XRD pattern of GO@CE (Figure 2b) shows a sharp peak at 8° and 11o and several sharp and broad peaks between 15° to 33o implying the presence of more crystals with various d-spacing. Also, the XRD pattern of [email protected] in Figure 2c indicate some sharp peaks and two broad peaks. The sharp peaks determine the crystallinity with various d-spacing while the broad peaks showed the nano-sized non-crystalline structures.
Morphology of samples was studied by FE-SEM images (Figure 3). FE-SEM image of GO@CE can be seen in Figure 3a which shows the graphene oxide sheets. FE-SEM image of [email protected] shows the graphene oxide sheets with more considerable distances due to its functionalization by tetramethylethylenediamine (Figure 3b).
EDAX analysis (Figure 4) of GO@CE confirms the presence of carbon, oxygen, and chlorine and their percentage in the table. EDAX analysis of [email protected] shows carbon, nitrogen, oxygen, and chlorine and their percent. Thus, EDAX analysis confirms the preparation of GO@CE and [email protected].
Thermogravimetric analysis (TGA) of [email protected] was conducted under argon atmosphere from 30 °C to 500 °C (Figure 5). TGA diagram shows two main weight losses. The first weight loss is around 100 °C, probably due to evaporation of water and low molecular weight solvents [48]. Second weight loss at 160 to 250 °C can be assigned to the isolation of functional groups [49]. The total weight loss and char yield are 27.24 and 72.76%, respectively. Therefore, the catalyst is stable under reaction conditions and can be used up to 200 °C in various reaction processes.

Figure 2. XRD pattern of a) GO, b) GO@CE and c) [email protected]

Figure 3. FE-SEM images of a) GO@CE and b) [email protected]

Figure 4. EDAX analysis of a) GO@CE and b) [email protected]
Synthesis of trisubstituted imidazoles using the catalyst
[email protected] was utilized to produce 2,4,5-trisubstituted imidazoles under solvent-free conditions via benzil, aldehydes, and ammonium acetate. The reaction of diketon, aldehyde, and ammonium acetate is the most used route for the production of trisubstituted imidazoles [50]. This route was studied using various catalysts. According to green chemistry principles, it suffers from some disadvantages [51]. Therefore, this work focuses on the removal or minimize disadvantages, mainly, environmental problems by employing a new graphene oxide based catalyst. In order to find optimum conditions, four variables such as catalyst, time, solvent, and temperature are examined for production of 3b (Scheme 2, Table 1). Optimized conditions for the production of imidazoles include 30 min., solvent-free conditions, 80 °C, 0.02 g of catalyst loading, and 95% yield (Table 1, entry 10).

Figure 5. TGA analysis of [email protected]
Table 1. Effect of catalyst ([email protected]) loading, temperature and time on the model reaction for the production of 3b

The capability of [email protected] catalyst was explored in the synthesis of 2,4,5-trisubstituted imidazoles from aldehydes (Scheme 2). Findings on the successful synthesis of 2,4,5-trisubstituted imidazoles are reported in Table 2 with a broad scope of functional groups, high yields, and sustainable conditions. Aldehydes with electron withdrawing-groups, and halogens offered higher yields of trisubstituted imidazoles compared to aldehydes with electron-donating substituents [52]. Electron-withdrawing groups accelerate nucleophilic attacks of ammonia on the carbonyl group by inducing a positive charge on the carbon of aldehyde. Moreover, they can facilitate water removal from the intermediate to attain final trisubstituted imidazole [53].
As mentioned in experimental section, final products were characterized by comparing their melting points with previously reported ones. Likewise, Nuclear magnetic resonance (NMR) spectroscopy confirmed the structure of 3k using DMSO-d6 as solvent and tetramethylsilane (TMS) as standard.

Scheme 2. Synthesis of 2,4,5-trisubstituted imidazoles catalyzed by [email protected] catalyst
Table 2. Synthesis of 2,4,5-trisubstituted imidazoles catalyzed by [email protected] catalyst

Table 3. Calculated values of turnover number (TON) and turnover frequency (TOF) for 2,4,5-trisubstituted imidazoles

To assess efficiency of [email protected] catalyst for production of trisubstituted imidazoles, turn over number (TON) and turn over frequency (TOF) values were calculated (Table 3). Turn over number (TON) was obtained using the equation: TON = product yield (g) / amount of catalyst (g) and turn over frequency (TOF) was afforded utilized the equation: TOF = product yield (g) / [reaction time (min) × amount of catalyst (g)] or TOF = TON / reaction time (min) [59].
Mechanism of reaction
A plausible mechanism of reaction is demonstrated in Figure 6. Ammonia was initially released from ammonium acetate and attacked to catalyst activated aldehyde to afford imine intermediate by releasing water (I), and then a second ammonia molecule attacked the imine to obtain II. In the next step, benzil (III) was attached by catalyst activated II via an adduct (IV) to achieve V. Ultimately, products were afforded by [1,5] hydride shift from intermediate V.

Figure 6. Plausible mechanism of reaction
Reusability and durability of catalyst
The recovery and reuse of the catalyst in the synthesis of 3b was explored for five runs to examine the stability of the catalyst under reaction conditions (in Figure 7) showed no vital changes in the catalyst recovery and reuse. Thus, the catalyst has sufficient stability and activity toward synthesizing 2,4,5-trisubstituted imidazoles.
Comparison with other reported catalysts
The efficiency of the reported catalyst was evaluated by the synthesis of 3e using the reported catalysts and comparing that with our catalyst (Table 4) in terms of reaction time, solvent, temperature, and catalyst loading, and this catalyst has priorities over previously reported ones.

Figure 7. Product yield of 3b and [email protected] catalyst recovery and reuse for five runs
Table 4. Comparison with previously reported catalysts

Conclusion
Graphene oxide functionalized tetramethylethylenediamine ammonium salt was prepared, characterized, and used as catalyst. Synthesis of 2,4,5-trisubstituted imidazoles was achieved successfully, using the catalyst, under solvent-free conditions via a one-pot reaction of benzil, ammonium acetate, and aldehydes. The developed catalyst offered several priorities over previously reported ones, including, nontoxic and carbon-based, recoverable under reaction conditions, easy handling and storage, and inexpensive. The reported process is sustainable and requires moderate conditions while offering high yields, and short reaction times. Considering the findings, the reported graphene oxide derivative can be employed in various fields in chemistry and material science.
Acknowledgments
The authors would like to appreciate the Payame Noor University (PNU) Research Council for supporting this research study.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Orcid
Esmael Rostami
https://orcid.org/0000-0002-6512-9951
How to cite this manuscript: Safie Mehrizi Marvast, Esmael Rostami. Graphene Oxide Modified with Tetramethylethylenediamine Ammonium Salt as a Powerful Catalyst for Production of Trisubstituted Imidazoles. Asian Journal of Green Chemistry, 8(3) 2024, 261-277. DOI: 10.48309/AJGC.2024.430848.1469