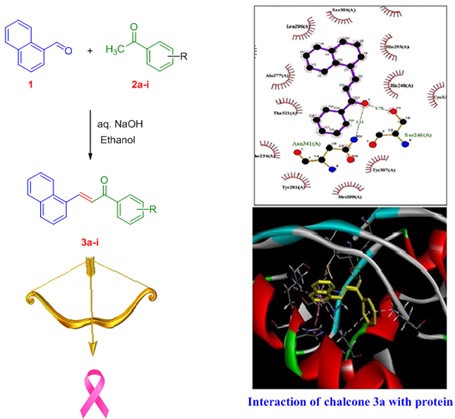
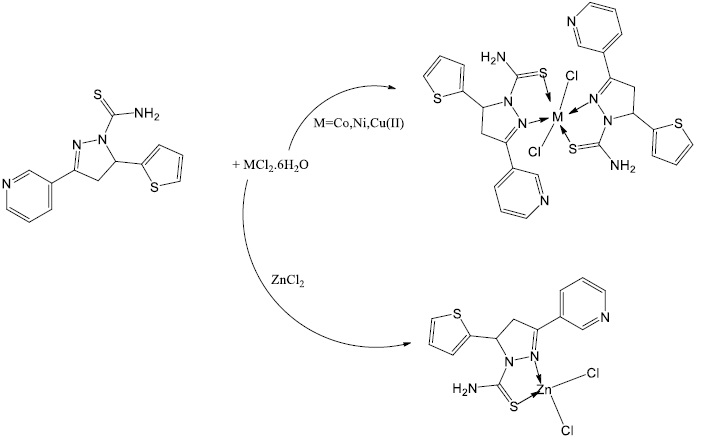
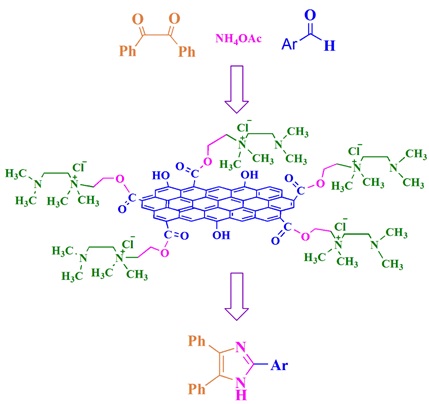
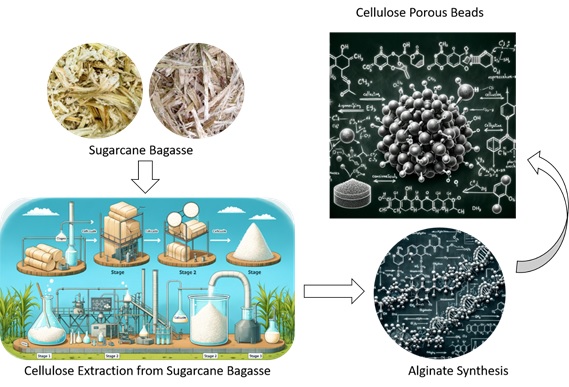
Burning or Burying Waste Increases the Risk of Cancer, Epidemics, and Environmental Issues Affecting the Atmosphere, Land, and Water
Pages 217-233
https://doi.org/10.48309/ajgc.2024.420730.1453
Salma Korbag, Issa Korbag
Abstract The growing population of Libya poses a significant environmental threat due to waste management challenges, including household, military, medical, electronic, hazardous metal landfills, heavy metal pollution, and methane emissions contributing to global warming. Waste management in Libya is poor, with incineration and landfilling leading to various diseases as cancer, respiratory, dermatological, and gastrointestinal ailments. Online surveys show a high rate of incineration (68%), followed by 23% landfilling, and while recycling rates are low or non-existent, with only 2%. Toxic metals can harm organs, water, soil, and air particles, necessitating waste management education and legislation on hazardous chemicals.