Document Type : Original Research Article
Authors
- Vijaykumar S. More 1
- Sharad P. Panchgalle 2
- Ranjit A. Gayake 3
- Vasant B. Jagrut 3
- Manisha M. Kodape 4
- Deekshaputra R. Birhade 5
- Mahendra N. N. Lokhande 6
1 Department of Chemistry, Kai. Rasika Mahavidyalaya Deoni, Dist. Latur, 413519, (M.S.), India
2 Department of Chemistry, K. M. C. College Khopoli, Dist. Raigad, 410203, (M.S.), India
3 Department of Chemistry, Swami Vivekanand Sr. College Mantha, Dist. Jalna, 431401(MS), India
4 Department of Chemistry, Sant Gadge Baba Amravati University, Amravati, 444602 (MS), India
5 Shri Vyankatesh Arts, Commerce and Science College Deulgaon Raja, Dist. Buldana, 443204 (MS), India
6 Department of Chemistry, Aavvaiyar Government College for Women Karaikal, Pondicherry, 609602, India
Abstract
Naphthalene chalcones (3a-i) with excellent yields were achieved through the use of aromatic ketones, 1-naphthaldehyde, and aqueous NaOH in the synthesis process. The synthesized chalcones were bio-evaluated as potential tubulin polymerization inhibitors for the treatment of breast cancer. The antiproliferative potential of each synthesized compound against the MCF-7 cell line was assessed. The majority of the compounds showed strong antiproliferative properties. With an IC50 value of 222.72 µg/mL, compound 3f exhibited the most antiproliferative activity among them, surpassing that of 5FU (IC50, 51.47 µg/mL).
Graphical Abstract
Keywords
Main Subjects
Introduction
Chalcones are important biologically active compounds [1, 2] and important synthons used to synthesize number of biologically active compounds (Scheme 1) as like pyrazolines, pyrrolyl, etc. Pyrazolines and derivatives [3] show biological activity as like anticonvulsant activity [4], antibacterial [5], anti-inflammatory [6], antifungal [7], antidepressant activity [8], antiamoebic [9], antituberculosis, antihyperglycemic, antimalarial, antileishmanial, and anticancer [10]. Six pyrrolyl derivatives show anti-inflammatory activity, anti-proteolytic activity, and also act as promising pleiotropic bioactive molecule [11].
Pyrazoline derivatives containing naphthalene exhibited the most anti-EGFR and anti-A549 human lung cancer cell line activities [12]. Guanidine derivatives act as potential antiproliferative and antitubulin polymerization inhibitors [13]. Ibrahim et al. showed path to find biologically active compounds by structure-based identification [14]. It is well known that introduction of heterocyclic moieties in compound leads to biological activity e.g., anticancer activity in pyrrolo[2,3-b]pyridine derivatives [15], thiohydantoin derivatives [16], pyrimidine derivatives [17], etc. Metal complexes of 2-hydroxychalcones had shown good antioxidant and antimicrobial activity [18].
Chalcones are versatile starting materials for many of heterocyclic compounds [19, 20]. Thus, chalcones are one of the most important compounds in medicinal chemistry and can be prepared using a simple but important reaction, i.e. Aldol condensation reaction.
The literature review reveals that chalcones have strong anticancer effects on various cancer cell lines [21-26]. By inhibiting tubulin polymerization, several artificial and natural chalcones shown strong anticancer efficacy against numerous cancer cell lines [22, 23, 27].
2-Hydroxy-4-methoxy-2',3'-benzochalcone (HymnPro, Scheme 2), which Shin and co-workers synthesized, has been shown to have antiproliferative activity in multiple human solid tumor cell lines and to inhibit the growth of xenografted tumors in nude mice [28]. The synthesis of 2'-hydroxy 5',6'-naphthochalcone derivatives was reported by Lee et al. [25]. Of these compounds, HMNC-74 (Scheme 2) has a potent inhibitory effect on the colon cancer cells SW620's ability to proliferate. Furthermore, it was shown that various substituents on the aromatic rings of chalcone might significantly alter the compounds' anticancer properties.
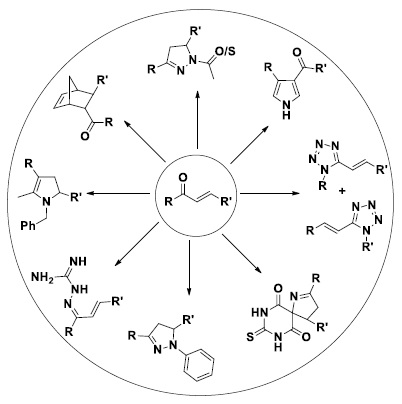
Scheme 1. Chalcones-source of bioactive compounds
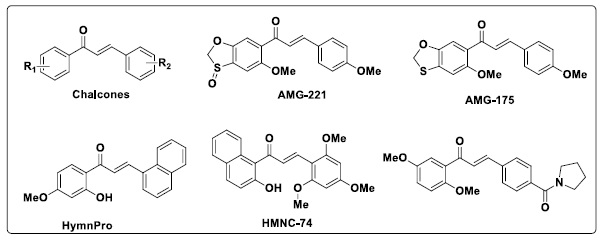
Scheme 2. Structures of some chalcones with anticancer activity
There is still plenty of room to create new chalcones with higher biological activity and investigate them, even if a number of chalcones have been synthesized and investigated for biological activities, including anticancer activity. Thus, we decided to synthesize naphthalene-chalcone derivatives and evaluate them for their anticancer activity.
Results and discussion
Chemistry
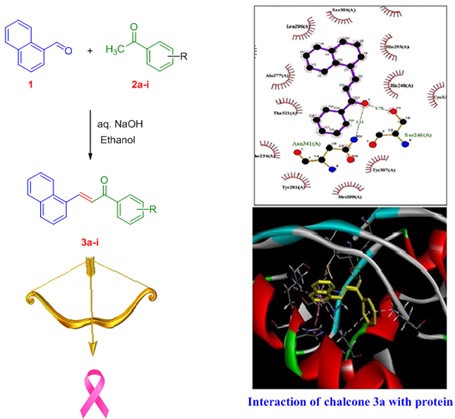
Naphthalene-chalcone derivatives 3a-i was prepared by condensing 1-Naphthaldehyde with different aromatic ketones using aqueous sodium hydroxide (Scheme 3). Structures of ketone and product chalcone were given in Table 1 along with useful information as like absorbance, concentration, % inhibition and IC50 values.
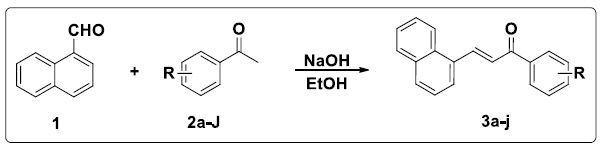
Scheme 3. Synthesis of naphthalene-chalcones 3a-i
To synthesize different substituted chalcones, 1-Naphthaldehyde (1eq.) was treated with different ketones (1 eq.) using NaOH (1 eq.) as base in ethanol. After completion of reaction (TLC monitoring), to obtain a crude solid material, ethanol was evaporated using a rotary evaporator at a lower pressure, and then small amount of water was added and extracted with ethyl acetate. Combined organic layer was dried over anhydrous Na2SO4 and evaporated to get crude product. The product obtained was further purified by recrystallization.
Biological activity
MTT assay
The in vitro tetrazolium-based colorimetric test (MTT) is a quick technique based on the way mitochondrial enzymes in metabolically active cells cleave a yellow tetrazolium salt into purple formazan crystals [29].
The antiproliferative activity of all freshly synthesized compounds 3a-i was assessed against MCF-7 cell lines of human breast cancer using the standard 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, with 5FU serving as the standard medication. The IC50 (µg/ml) values (half maximal inhibitory concentration) of the tested compounds 3a-i and the standard drug 5FU are listed in Table 1.
Experimental procedure
(1) The cells were cultured in culture media for 24 hours at 37 °C and 5% CO2 at a concentration of 1×104 cells/mL.
(2) In micro plates (tissue culture grade, with 96 wells), cells were seeded at a density of 104 cells/well in 100 µl culture media and 100 µl of sample 3a-j at a concentration of 1000µg/mL, respectively.
(3) Cell line and DMSO (0.2% in PBS) were cultured in control wells. Each sample underwent three rounds of incubation.
Table 1. Chalcones (3a-i) with concentration, absorbance, % inhibition, and IC50 values

To find the proportion of live cells after culture and the survival rate of control cells, controls were kept in place.
(4) Cell cultures were placed in a CO2 incubator (Thermo Scientific BB150) and incubated for 24 hours at 37 °C with 5% CO2.
(5) Following the incubation period, the medium was fully extracted and 20 µl of MTT reagent (5 mg/min PBS) was added.
(6) Cells were cultured for 4 hours at 37 oC in a CO2 incubator following the injection of MTT.
(7) With a microscope, examined the wells for formazan crystal development. Only live cells were able to convert the yellowish MTT to a dark-colored formazan.
(8) Completely removed the medium, added 200 µl of DMSO, let it sit for 10 minutes, and then incubated (covered in aluminum foil) at 37 oC.
(9) The absorbance of each of the three samples was measured using a microplate reader (Benesphera E21) set to operate at 550 nm.
Based on the acquired data, it was noted that the majority of the compounds that were synthesized demonstrated strong antiproliferative activity. At concentration 100 µg/mL, chalcones 3a, 3e, and 3f showed good percent inhibition MCF-7 cell line, as compared to standard drug. The compounds 3a, 3e, and 3f showed lowest IC50 value as compared to other compounds. It indicates these compounds showed 50% inhibition of cell growth at this concentration. It was observed that the presence of group that donate electrons on phenyl ring causes the inhibitory action to slightly increase. It was discovered that compound 3f (IC50 = 222.72 μg/mL), which has methoxy at the phenyl ring's 2-position, is the most active of the group. When compared to the non-substituted ring itself, the phenyl ring's 3-methoxy substituent exhibits lesser activity and the methoxy substituent at positions 2 and 4 exhibits higher activity. Electron withdrawing substituent like –NO2, -Cl, and –Br group show decreased activity. 4-Br compound 3b shows lowest activity with IC50 value 818.18 μg/ml while 4-Cl compound 3c shows IC50 value 498.77 μg/mL. 4-NO2 and 3-NO2 compounds show IC50 value 383.82 and 465.60 μg/mL, respectively, which are more as compared to methoxy substituent.

Figure 1. Graph showing IC50 values of different chalcones
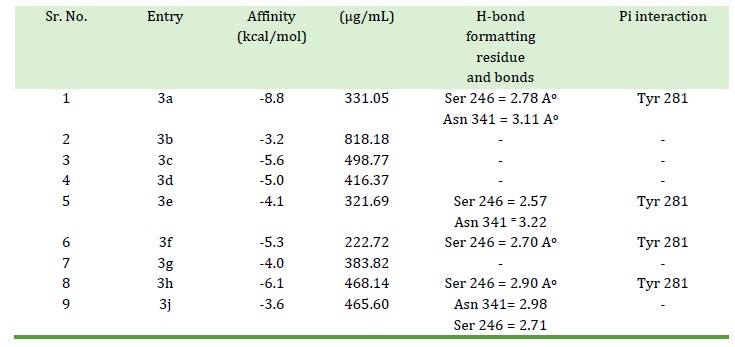
Table 2. Docking score interaction affinity for all chalcone derivatives
This indicates that electron donating substituent at 2-position increases the activity of the synthesized chalcones.
This can be cleared from the graph plotted between chalcones and corresponding IC50 values (Figure 1).
Molecular docking studies
Preparation of protein and ligand
The protein, whose crystal structure features a high affinity hetero dimer of the ARNT C-terminal PAS domain and HIF2α, was downloaded from the RCSB (www.rcsb.org) Protein Data Bank (PDB ID: 3F1O).
Using discovery studio to prepare the PDB format for the docking investigation, the interaction between the substituted chalcone derivatives and the aforementioned protein was investigated. The auto dock 4.2 software tool carried out the computations for molecular docking [30]. After preparing the compounds' molecular structures and minimizing their energy, the compound with the lowest free binding energy was selected for additional examination.
Docking analysis
The 3D crystal structure of HIF2α (PDBID: 3F1O) was used for this docking study. The Auto Dock Tools were used to eliminate every unattached water atom and molecule. The protein was then treated to the addition of polar hydrogen atoms and Kollman-Gesteiger charges.
We used the Lamarckian Genetic Algorithm (LGA) for all docking computations [31]. The grid box was placed at center with coordinates x: 12.378650, y: -41.613200, z: 10.343900. Docking was carried out under the same circumstances and with the same Auto Dock parameters for each compound.
Each compound's lowest binding energy conformation was chosen following the completion of the docking investigation. After comparing the propensity of each chalcone derivative to interact with protein, it was discovered that compound 3a exhibits good inhibition (having the highest interaction affinity). Docking score interaction affinity for all chalcone derivatives are presented in Table 2 along with useful information as like H-bond formatting residue, pi interactions, etc.
Compound 3a shows IC50 value of 331.05 µg/mL and in docking study shows highest affinity for protein (-8.8 kcal/mol). LigPlot [32] software shows hydrogen bonding interaction with Ser 246 (A) and Asn 341 (A) at distance of 2.78 and 3.11 Ao, respectively, depicted in Figure 2 and Figure 3. There are pi-interactions with Tyr 281(A) and hydrophobic interactions with other amino acids.
Compounds 3h shows IC50 value of 468.14 µg/ml and in docking study indicates the highest affinity for protein (-6.1 kcal/mol). LigPlot software shows hydrogen bonding interaction for 3h with Ser 246 A at distance of 2.90 Ao. Compound 3h also shows pi-interactions with Tyr 281(A) and hydrophobic interactions with other amino acids.
Compound 3f shows good IC50 value (222.72 µg/ml) and in docking study Affinity for protein (-5.3 kcal/mol). LigPlot software shows interaction with Ser 246 (A) at distance of 2.70 Ao and pi-interactions with Tyr 281(A). It also reveals hydrophobic interaction with other amino acids.
IC50 value of 321.69 µg/mL was obtained for compound 3e and in docking study affinity for protein (-4.1 kcal/mol). LigPlot software shows hydrogen bonding interaction with Ser 246 (A) and Asn 341 (A) at distance of 2.57 and 3.22 Ao, respectively. It also shows pi-interactions with Tyr 281 (A) and hydrophobic interactions with other amino acids.
The correlation value between the theoretical and experimental data is provided by the Pearson correlation coefficient (R2). Pearson's correlation coefficient shows a linear relationship between two variables that spans from +1 to -1. A complete positive linear relationship between variables is shown by a correlation value of +1. Predicted binding free energies and actual IC50 values against MCF-7 human breast cancer cells are correlated. The results indicate a positive association between affinity (kcal/mol) and IC50 (µg/mL), as indicated by the computed Pearson correlation coefficient of 0.423756 for this study.
Experimental
General experimental procedure
After dissolving the starting ketone 2 (1eq.) in ethanol, 1eq. of sodium hydroxide was added. After agitating the reaction mixture for ten minutes at room temperature, one equivalent of 1-naphthaldehyde was added.
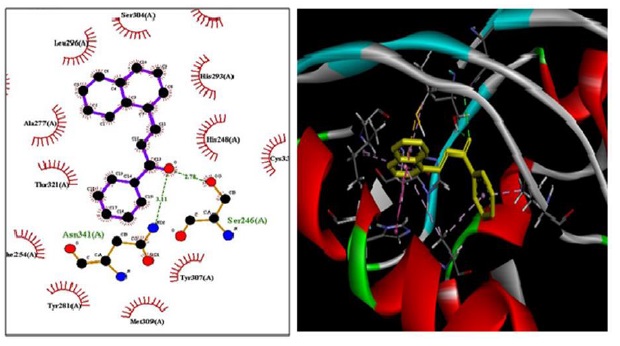
Figure 2. Interaction of compound 3a with protein
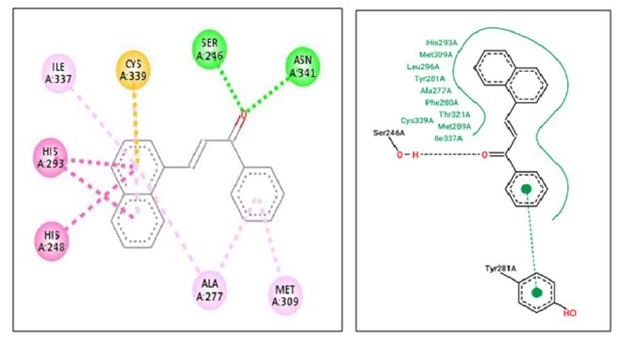
Figure 3. Compound 3a showing 2D interaction with protein
For 30 minutes, the reaction mixture was continuously stirred. The progress of reaction was monitored by TLC analysis. Crude product was obtained by removing the solvent on a rotary evaporator under decreased pressure after the reaction was completed (TLC check). To obtain the desired product 3, water was added to the crude product and it was extracted three times using ethyl acetate. Recrystallization was used to further purify the product.
(E)-3-(naphthalen-1-yl)-1-phenylprop-2-en-1-one (3a)
IR (KBr) (νmax/ cm-1): 3016, 1695, 1636, 1449, 1215, 744, and 666. 1H NMR (400 MHz, CDCl3): δ 8.40 (d, J = 15.8 Hz, 1H), 8.12 (d, J = 6.9 Hz, 1H), 7.95-7.87 (m, 4H), 7.67-7.40 (m, 7H), and 7.29 (d, J = 4.2 Hz, 1H). 13C NMR (101 MHz, CDCl3): δ 193.54, 142.93, 139.26, 136.66, 135.31, 133.76, 131.61, 131.18, 130.49, 129.58, 129.09, 128.87, 128.56, 126.98, 126.36, 125.50, 124.90, and 123.2. GC MASS= 258
(E)-1-(4-bromophenyl)-3-(naphthalen-1-yl)prop-2-en-1-one (3b)
IR (KBr) (νmax/ cm-1): 3059, 1659, 1593, and 590. 1H NMR (400 MHz, CDCl3): δ 8.60 (d, J = 15.6 Hz, 1H), 8.24 (d, J = 8.2 Hz, 1H), 8.03-7.93 (m, 3H), 7.71-7.45 (m, 6H), and 7.18 (m, 2H). 13C NMR (101 MHz, CDCl3): δ 189, 142, 140.1, 136.5, 136.02, 134.5, 132.01, 132, 131, 131.5, 130.02, 126.7, 126, 125.51, 125.0, 124.05, and 123. LC MASS= 351.5 (M+ Na).
(E)-1-(4-chlorophenyl)-3-(naphthalen-1-yl)prop-2-en-1-one (3c)
IR (KBr) (νmax/ cm-1): 3060, 1656, 1590, and 592 cm-1. 1H NMR (400 MHz, CDCl3): δ 8.49 (t, J = 18.5 Hz, 1H), 8.23 (t, J = 14.6 Hz, 1H), 8.01-7.81 (m, 3H), 7.74-7.63 (m, 2H), 7.66-7.50 (m, 12H), 7.40-7.23 (m, 5H), and 7.25-7.00 (m, 6H). 13C NMR (101 MHz, CDCl3): δ 166.37, 158.30, 151.81, 140.06, 137.15, 133.16, 130.28, 122.54, 127.50, 124.01, 125.34, 121.02, 122.30, 119.04, 111.72, and 55.80. GC MASS= 292.
(E)-1-(3-methoxyphenyl)-3-(naphthalen-1-yl)prop-2-en-1-one (3d)
IR (KBr) (νmax/ cm-1): 2925, 1652, 1592, 1437, 1211, 771, and 665. 1H NMR (400 MHz, CDCl3): δ 8.52 (d, J = 15.6 Hz, 1H), 8.25 (t, J = 9.4 Hz, 1H), 7.89 (dt, J = 18.1, 9.5 Hz, 3H), 7.75 (d, J = 7.6 Hz, 1H), 7.66-7.47 (m, 5H), 7.11 (t, J = 7.5 Hz, 1H), 7.05 (d, J = 8.4 Hz, 1H), 3.95 (s, 3H). 13C NMR (101 MHz, CDCl3): δ 192.69, 158.29, 139.92, 133.76, 133.11, 132.64, 131.79, 130.51, 129.70, 129.34, 128.73, 126.81, 126.21, 125.59, 125.05, 123.61, 120.85, 111.71, and 55.79. GC MASS= 289.
(E)-1-(4-methoxyphenyl)-3-(naphthalen-1-yl)prop-2-en-1-one (3e)
IR (KBr) (νmax/ cm-1): 2920, 1648, 1595, 1435, 1215, 770, and 660. 1H NMR (400 MHz, CDCl3): δ 8.54 (d, J = 15.6 Hz, 1H), 8.26 (d, J = 8.2 Hz, 1H), 7.99-7.83 (m, 3H), 7.75 (dt, J = 13.7, 6.8 Hz, 1H), 7.55 (ddd, J = 15.6, 12.7, 4.2 Hz, 5H), 7.11 (t, J = 7.3 Hz, 1H), 7.03 (t, J = 9.3 Hz, 1H), and 3.93 (d, J = 9.9 Hz, 3H). 13C-NMR (101 MHz, CDCl3): δ 192.71, 158.30, 139.91, 133.77, 133.13, 132.60, 131.79, 130.42, 129.68, 129.31, 128.76, 126.84, 126.23, 125.51, 125.17, 123.60, 120.86, 111.73, 111.73, 55.79, and 55.46. GCMS= 289
(E)-1-(2-methoxyphenyl)-3-(naphthalen-1-yl)prop-2-en-1-one (3f)
IR (KBr) (νmax/ cm-1): 2923, 1654, 1590, 1440, 1219, 770, and 663. 1H NMR (400 MHz, CDCl3): δ 8.68 (d, J = 15.4 Hz, 1H), 8.30 (d, J = 8.3 Hz, 1H), 8.12 (d, J = 8.9 Hz, 2H), 7.98-7.88 (m, 3H), 7.69-7.51 (m, 4H), 7.03 (d, J = 8.8 Hz, 2H), and 3.92 (s, 3H). 13C NMR (101 MHz, CDCl3): δ 188.52, 163.52, 140.92, 133.76, 132.67, 131.79, 131.10, 130.92, 130.58, 128.73, 126.91, 126.27, 125.45, 124.99, 124.66, 123.61, 113.93, and 55.48. GC MASS= 289
(E)-3-(naphthalen-1-yl)-1-(4-nitrophenyl)prop-2-en-1-one (3g)
IR (KBr) (νmax/ cm-1): 3020, 1679, 1519, 1215, and 742. 1H NMR (400 MHz, CDCl3): δ 8.68 (m, 2H), 8.31 (m, 2H), 8.1-7.8 (m, 3H), 7.65-7.37 (m, 4H), and 7.27 (m, 2H). 13C NMR (101 MHz, CDCl3): δ 188.95, 140.25, 139.95, 136, 130.4, 133.1, 131, 130, 129, 126, 125, and 124. LC MASS= 325 (M+ Na).
(E)-1-(2-chlorophenyl)-3-(naphthalen-1-yl)prop-2-en-1-one (3h)
IR (KBr) (νmax/ cm-1): 3061, 1650, 1592, and 595. 1H NMR (400 MHz, CDCl3): δ 8.40 (d, J = 14.5 Hz, 1H), 8.04 (d, J = 8.3 Hz, 1H), 8.01-7.85 (m, 4H), 7.70-7.40 (m, 5H), 7.25 (m, 2H). 13C NMR (101 MHz, CDCl3): δ 189, 142, 139.6, 136, 134, 132, 131, 130.50, 130, 129, 126.50, 125, 124, and 123. LC MASS= 314.8 (M+ Na).
(E)-3-(naphthalen-1-yl)-1-(3-nitrophenyl)prop-2-en-1-one (3i)
IR (KBr) (νmax/ cm-1): 3025, 1672, 1522, 1212, and 740. 1H NMR (400 MHz, CDCl3): δ 8.68 (m, 2H), 8.25 (m, 3H), 8.01(m, 4H), 7.51 (m, 4H). 13C NMR (101 MHz, CDCl3): δ 188.95, 142.21, 139.34, 136.51, 134, 132, 131.5, 131, 130, 128.50, 126, 125.1, 124, and 123.61. LC MASS= 325.6 (M+ Na).
Conclusion
To sum up, we designed and synthesized a range of naphthalene-chalcone derivatives (3a-j) and evaluated their efficacy in treating MCF-7 cell line-derived breast cancer. With an IC50 value of 222.70 µg/mL, compound 3f, which carries a 2-methoxy phenyl moiety, is the most potent molecule against the MCF-7 breast cancer cell line among them. Moreover, molecular docking analyses demonstrated that these compounds show affinity for proteins.
Acknowledgements
Savitribai Phule Pune University, Pune is acknowledged by the authors for its analytical support. No specific grant was given for this research by public, private, or nonprofit funding organizations.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Orcid
Vijaykumar S. More
https://orcid.org/0000-0002-6110-8774
Sharad P. Panchgalle
https://orcid.org/0000-0001-9706-7567
Ranjit A. Gayake
https://orcid.org/0000-0003-2760-9601
Vasant B. Jagrut
https://orcid.org/0000-0003-2760-9601
Manisha M. Kodape
https://orcid.org/0000-0001-6185-9001
Deekshaputra R. Birhade
https://orcid.org/0000-0003-4007-0057
Mahendra N. Lokhande
https://orcid.org/0000-0002-9226-119X
How to cite this manuscript: Vijaykumar S. More, Sharad P. Panchgalle, Ranjit A. Gayake, Vasant B. Jagrut, Manisha M. Kodape, Deekshaputra R. Birhade, Mahendra N. Lokhande*. Synthesis, Molecular Docking, and Biological Evaluation of Some New Naphthalene-Chalcone Derivatives as Potential Anticancer Agent on MCF-7 Cell Line by MTT Assay. Asian Journal of Green Chemistry, 8(3) 2024, 234-246. DOI: 10.48309/AJGC.2024.418444.1458