Document Type : Original Research Article
Authors
1 Department of Chemistry, College of Sciences for Girls, University of Babylon, Hilla, Iraq
2 Department of Medical Laboratories Technology, Al-Manara College for Medical Sciences, Maysan, Iraq
3 Department of Medical Laboratories Technology, Al-Hadi University College, Baghdad,10011, Iraq
4 Department of Medical Laboratories Technology, AL-Nisour University College, Baghdad, Iraq
Abstract
This study demonstrates peanut shells as a potential inexpensive and ecofriendly adsorbent for adsorption of streptomycin drug from aqueous solution via batch adsorption method. Reaction isotherm studies were carried out at several experimental conditions of concentration of streptomycin drug (10-100 mg/g), adsorbent dosage (0.1-2 g), equilibrium time (5-60 min), and solution pH (3-10). The adsorbent surface was characterized via field emission scanning electron microscopes (FE-SEM) and transmission electron microscopy (TEM). The best adsorption efficiency of drug on to peanut shells was observed as high as 1.677 mg/g. The removal percentage % of streptomycin drug rise with increasing weight of peanut shells as well as solution of pH. Equilibrium of streptomycin drug on to peanut shells rind is found to be quick, and equilibrium to reached in 60 min. Langmuir isotherm and Freundlich isotherm models were useful to fit the result of equilibrium peanut shells, and it was noticed that isotherm Freundlich fits the multilayer peanut shells ability for drug was 1.455 mg/g. These data demonstrated that peanut shells are locally available, ecologically, effective and inexpensive adsorbent for the adsorption of Streptomycin drug from aqueous solution.
Graphical Abstract
Keywords
Main Subjects
Introduction
Emerging pollutants refer to artificially created substances that have been found in surface water in trace quantities [1]. Aquatic ecosystems are accessed through different routes such as the wastewater discharge from medical clinics or pharmaceutical manufacturing areas, as well as the animal waste originating from poultry farms, aquaculture facilities, and more [2].
Over the past years, the increase in population growth has led to the increase and development of industries in developed countries, and this leads to an increase in environmental problems due to industrial liquid waste. Tons of pollutants and chemical compounds are discharged into wastewater without adequate treatment. Among these toxic pollutants, they include pharmaceutical that are widely used in industries. In general, the presence of pharmaceuticals contaminants in the aquatic environment can be observed in very low concentrations [1, 2]. However, they are considered among the green pollutants because they are not easily distinguished and due to their inability to color. They also affect the aesthetic aspect of the environment and reduce the penetration of light, which reduces oxygen levels, and thus affects the system. Environment for animals and plants. The presence of drugs in very low concentrations in water sources is associated with severe harmful effects on animal and human health [3-6].
Agricultural wastes like coconut shell, date stone, bunch waste, jujube seed, peanut shell, cashew nut peel, cocoa pod shell, sugar cane bagasse, rice husk, neem leaf, periwinkle peel, garlic shell, peanut shells, eggshell, bagasse pith, olive waste cake, peanut husk wastes apple, rice shell, waste tea, orange shell, coir pith, corn cobs, and barley husk neem leaves, wheat bran, and peanut hulls, banana shell, Bengal gram seed shell, oil palm fruit waste, castor bean shell, maize stem, and lignite have been used as inexpensive alternatives as well as low-cost adsorbents in the uptake of drug [7-11].
Streptomycin is an antibiotic medication and the first discovered aminoglycoside, isolated from Streptomyces bacteria, was utilized for treatment of a number of bacterial infections, like Mycobacterium valium complex, pulmonary tuberculosis, plague, brucellosis, tularemia, endocarditis, Burkholderia infection, and mouse bite fever [12, 13]. Chemical formula was C21H39N7O12, molar mass was 581.580 g·mol-1, and chemical stretcher is shown in Scheme 1.
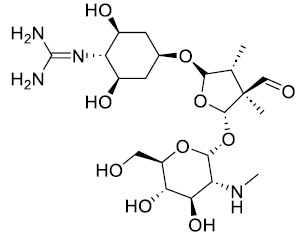
Scheme 1. Chemical structure of streptomycin drug
Experimental
Preparation of adsorbent
The peanut shells were obtained from local market in Iraq. The peanut shells were separated from the edible pulp, and washed well with water to remove unwanted dust and suspended contaminations. They were cut into small pieces and dried in the sunlight for three days until a stable mass was obtained. The peanut shells were ground, and dried in a special mill, then sieved to obtain particles with a diameter of less than 50 nm, and stored in tightly sealed glass bottles and used in all experiments without any chemical or physical treatment.
Adsorption studies of several parameters
Optimization of factors in the study including the investigation in to equilibrium time required to reach stability (5-60 min), pH solution (3-10), and concentration of Streptomycin drug (10-100 mg/L) and weight of peanut shells (0.1-2 g). Adjustment of solution pH was done utilizing NaOH or HCl (0.1 M). Adsorption equilibrium isotherm was carried out in concentration (50 mg /L). All investigates in this study were carried out with RDFP (0.020 g) in solution drug 100 ml and suspensions was shaken for 60 min at 250 rpm at 25 oC. The removal percentage % and adsorption capacity calculate in Equations (1) and (2).
Figure 1 displays FE-SEM of bean peels. The surface is rough with large aggregates on the surface and has a fiber-like structure, which is a distinctive characteristic of agricultural waste (Figure 1a). However, the surface roughness has different apertures. The sizes help to increase the number of effective sites on the surface as a result of the surface containing large pores, and these openings can serve as transport pores for pollutant particles. After the adsorption process, it was observed that the pores on the surface were filled so that the surface of peanut shells was covered by the adsorbed molecules (Figure 1b). To determine the shape, size and particle distribution of tea waste, TEM analysis was performed. As depicted in Figure 1c, dark-colored semi-spherical aggregates enclosed within a matrix-like structure were obtained, which can be attributed to the process of activation and treatment with acid. The process of activation increases the surface porosity and also the active aggregates on the surface of the preparation [14, 15].

Figure 1. FE-SEM image a) peanut shells before adsorption, b) peanut shells after adsorption, and c) TEM of bean peels
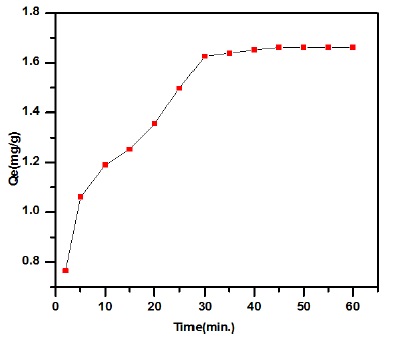
Figure 2. Effect of equilibrium time onto adsorption capacity streptomycin drug by bean peels
Effect of contact time
Figures 2 illustrate the interactions of adsorbate-adsorbent as a parameter of time at concentration (20 mg/L). The uptake of drug via peanut shells was observed to be quick; the amount adsorption capacity (Qe mg/g) increased with rise time, equilibrium was reached at 1 h. Though, concentration of drug 20 mg/L constant was reached after 1 h. The maximum adsorption capacity at equilibrium rise (1.662 mg/g) for 1 h. Based on the data, 1 h was selected as contact time bean peels. Initially, this rapid increase is a result of the high absorption of pollutant molecules to the active sites on the surface. After that, the increase is slow as a result of most of the surfactant sites being saturated by the adsorption process with drug molecules.
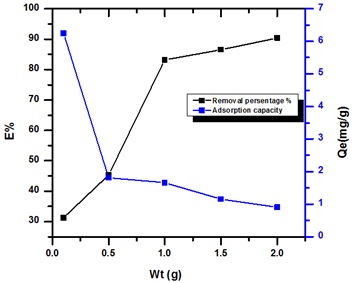
Effect weight of peanut shells
The influence dosage of peanut shells on adsorption drug was studied in different weight from (0.1-2 g), as shows in Figure 3. The removal of adsorbed drug, increases with rise in weight from (31.22-90.87%), but adsorption capacity (Qe mg/g) decrease when increase of weight from (6.266-0.988 mg/g). The point of saturation all active site for Peanut shells at 1 g for the percentage removal drug (83.14%) and point saturation adsorption capacity at 1.662 mg/g for drug.
There was no important drug removal observed above the point saturation due to the establishment of equilibrium among the molecules drug on the adsorbent and in the solution. Based on the results, the adsorption efficiency showed a completely opposite trend to the removal percentage, where it decreased with increasing weight of PG/BCC. The reason may be due to an increase in the adsorbent weight at a fixed concentration, which causes most of the active sites to remain unsaturated [16, 17].
Effect of solution pH
To study the influence of pH solution on uptake equilibrium capacity of bean peels, at concentration of drug 20 mg/L, weight of peanut shells 1 g and 25 ᵒC. Figure 4 demonstrates that the drug sorption was minimum at pH 3 (1.111 mg/g), (55.60 %) and increased adsorption capacity with pH increase up to 6 (1.45 mg/g), (83.87 %) and then reach constant at pH ranges of 8-10. The adsorption capacity and removal percentage rises with increase in solution pH (1.62-1.66 mg/g), (81.22-83.65%) and the maximum removal in basic medium. Thus, the electrostatic repulsion forces between drug cations and the protonated sites of PG/BCC resulted in decreased adsorption efficiency. Moreover, decreased drug removal in acidic media could be due to the occurrence of competitive adsorption between drug cations and protons. High drug removal in basic media was due to electrostatic attraction between the negatively charged adsorbent surface and drug cations [18-20].
Effect of concentration drug
The primary concentration of drug provides a significant parameter between solid phases and aqueous. The influence of the primary concentration of drug on the adsorption efficiency of peanut shells was estimated at several concentrations: 10-100 mg/L. The data showed that with the increase in concentration of the drug from 10-100 mg/L, the adsorption efficiency of pean shells increased from 0.987-3.988 mg/g [5,21], but removal percentage % decrease from 65.66-99.76 % illustrated in Figure 5.
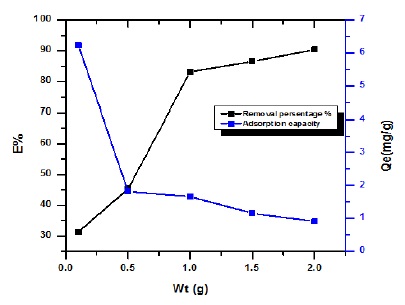
Figure 3. Effect of weight of peanut shells onto removal Streptomycin drug
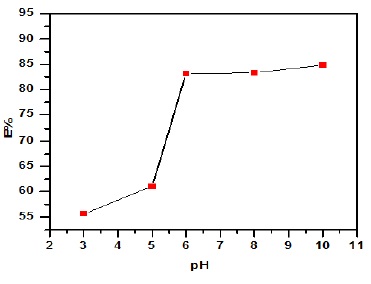
Figure 4. Effect of pH solution onto removal streptomycin drug
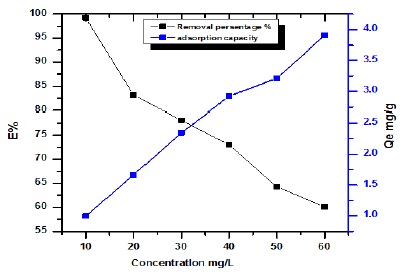
Figure 5. Effect of concentration streptomycin drug onto adsorption capacity
Adsorption isotherm
The adsorption process, which distinguishes between the liquid and solid phases, is considered a dynamic equilibrium process. Therefore, the isotherm process can explain the mechanism of distribution of adsorbed molecules between the liquid and solid phases in an equilibrium state, which provides practical advantages for the equilibrium isotherm to give results about the surface properties with the affinity of the used adsorbent and the mechanism of adsorption process to improve the removal process [22]. Freundlich’ isotherm and Langmuir isotherm, the most widely utilized adsorption models, were utilized to fit the experimental equilibrium result and gain insight into the adsorbate’s bio-sorption behavior in the adsorbent presence. The model Langmuir isotherm assumes the existence of an adsorption monolayer on a site homogeneous distribution over the adsorbent surface without interaction with adsorbate. In contrast, the adsorption multilayer occurrence on heterogeneous surface assumption is the basis of the model Freundlich isotherm [23, 24]. Freundlich, Langmuir in their nonlinear calculation of Equations (3) and (4).

The values of R2 and KF are found in the Freundlich model, as depicted in Figure 6. The adsorption of Streptomycin is the drug best fitted in the Freundlich model with the better R2= 0.8855 based on the comparison with model Langmuir. The result found that the model Freundlich exhibited a better fit to the adsorption result than the model Langmuir isotherm. All parameters of models are indicated in Table 1.
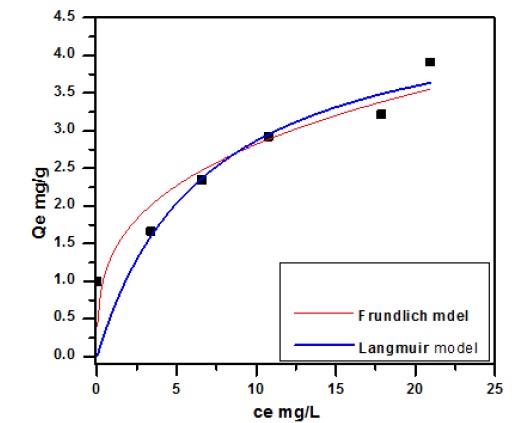
Figure 6. Adsorption models nonlinear fit of adsorption streptomycin drug onto bean peels, con. 20 mg. L-1, mass 1 g, and temperature = 25 °C
Table 1. Isotherm Langmuir and Freundlich parameters streptomycin drug adsorbed onto bean peels

Conclusion
The data established that the bio-sorbent prepared from peanut shells, a low-cost agricultural waste, is promising for drug removal from aqueous solutions because of its obtainability as waste and ecological adsorbents. The operating parameters, pH of the solution of the drug, equilibrium time, weight of peanut shells, and concentration of the drug were effective in drug removal utilizing peanut shells in batch experiments. The equilibrium result was better simulated on the isotherm Freundlich model with the best adsorption capacity of 1.445 mg/g. The adsorption capacity and percentage of removal of drugs increased with rising contact time and surface area. But adsorption capacity (Qe mg/g) decreased with the weight increase. The maximum contact time for equilibrium to be reached is 60 min. The removal percentage of the drug was to increase with an increase in the pH solution in the alkane medium.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the article and agreed to be responsible for all the aspects of this work.
Orcid
Ayad F Alkaim
https://orcid.org/0000-0003-3459-4583
Aseel M. Aljeboree
https://orcid.org/0000-0001-5397-3330
Ola Hamad Salah
https://orcid.org/0000-0002-3194-1749
Usama S. Altimari
https://orcid.org/0000-0002-7485-5875
How to cite this manuscript: Ayad F Alkaim *, Aseel M. Aljeboree, Ola Hamad Salah, Ammar Ali Aljanabi,Usama S. Altimari. Adsorption of Toxic Drug Pollutants from Aqueous Solutions onto Biosorbent: Nonlinear Regression Analyses of Isotherm Models. Asian Journal of Green Chemistry, 8(2) 2024, 188-197. DOI: 10.48309/ajgc.2024.427437.1467