Document Type : Original Research Article
Authors
1 Department of Chemistry , Faculty of Science , University of Benghazi, Benghazi, Libya
2 Libyan International Medical University, Faculty of Applied Medical Science, Department of Biological Sciences
3 Department of Chemistry, Faculty of Arts & Science, University of Benghazi, Alabyar, Libya
Abstract
The nutritional value of honey depends on its chemical composition, which is influenced by the surrounding environment such as the botanical source and geographical origin. In a study, these twenty samples of honey were collected from various regions in Libya and analyzed to illustrate this variation. During our study, we discovered that all honey samples were contaminated with trace elements, which can be attributed to various factors such as the use of steel or galvanized containers in processing, shipping, or storage, exposure to insecticides, and human activity. As a result, the interpretation of the results may not be entirely reliable due to the presence of various unknown variables. Electrothermal atomic absorption spectrometry was utilized to analyze the tree trace elements Ni, Cd, and Pb. Furthermore, a statistical analysis was conducted to determine any existing correlation between the geographical origin and the variation in the presence of these elements from one sample to another. In addition, the pollen grains analysis of all honey samples was performed to identify the botanical source of each sample and interpret the variations in the nutritional value of the elements content from one sample to another. The ranges of the elements were as follows: Ni (0.20-0.75 mg/kg), Cd (0.250-1.75 mg/kg), and Pb (0.25-51.00 mg/kg).
Graphical Abstract
Keywords
Main Subjects
Introduction
Honey is commonly produced by bees using nectar extracted from flowers. It is a widely known sweetener that has been used across the world since ancient times. In addition to being a natural sweetener, it has also been used as a remedial agent [1, 2].
While there is a wealth of data available on the properties of honey from regions such as North America, Europe, Australia, India, and South Africa, information on Libyan honey is scarce. To address this gap, our paper outlines our initial findings from analyzing honey samples collected from sixteen locations in the east, two locations in the west, and two additional locations in the southwest regions of Libya.
Beekeeping is often undertaken in locations situated in or near residential and industrial areas.
However, if bee colonies are placed in unsuitable or inadequate apiaries, or if incorrect production methods are employed, this can adversely affect the quality and natural characteristics of bee products. In addition, environmental problems that result from population growth, urbanization, industrialization, and changing consumption patterns are becoming more prevalent.
For instance, plants can accumulate heavy metals from dust and ash emissions released into the air from various sources [3].
Bees come into contact with metal pollutants during their flights and while collecting water, pollen, and nectar as food resources. These metals accumulate in the bodies of bees and in the hive products they produce, which can have negative effects on their survival, physiology, and behavior [4].
The primary sources of heavy metals in agricultural soil are inorganic and organic fertilizers, which are substances added to the soil to improve plant growth and yield, along with sources such as liming, sewage sludge, irrigation waters, and pesticides [5].
Heavy metal toxicity is a cause for environmental worries in regions where mining, industry, and agriculture processes are present. This is because, when present in high concentrations, heavy metals can be fatal to honey bees.
However, it remains unclear how sublethal doses of heavy metal contamination may affect bees or if bees will consume food that is contaminated [6].
Experimental
Materilas and Methods
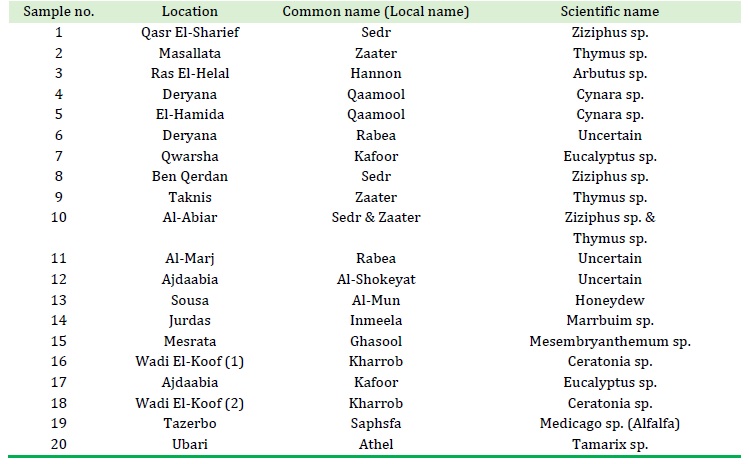
The samples were collected from different areas in Libya (Tables 1 and 2). East regions (Elqwarsha, Ras Elhelal, Sousa, Wadi Alkoof, Jurds, Elhemida, Taknis, Deryana, Elabiar, Benqerdan, Elmari, and Ejdabia) samples 2-16; west regions (Qasr Elshrief, Masallata, and Mesrata) samples 1, 17, and 18; southeast region (Tazerbo) samples 19 and 20; and southwest region (Aobari).
About 1 g of the representative sample was measured by using a sensitive analytical balance and transferred to a 25 mL volumetric flask, heated in a water bath to decrease the viscosity 0.25 mL nitric acid and 2.5 mL concentrated hydrogen peroxide were added, followed by 0.1 mL of ammonium dihydrogen phosphate to those samples being analyzed tree elements (Ni, Cd, and Pb). Then samples were diluted with 25 mL of deionized water and were sonicated for 5 minutes with continuous stirring. Finally, the samples were analyzed using the graphite furnace micro atomic absorption spectrometry [7]. In Micro Analytical Center, Faculty of Science, Cairo University.
Table 1. Name of Location where honey samples were collected and surrounding nature of hives
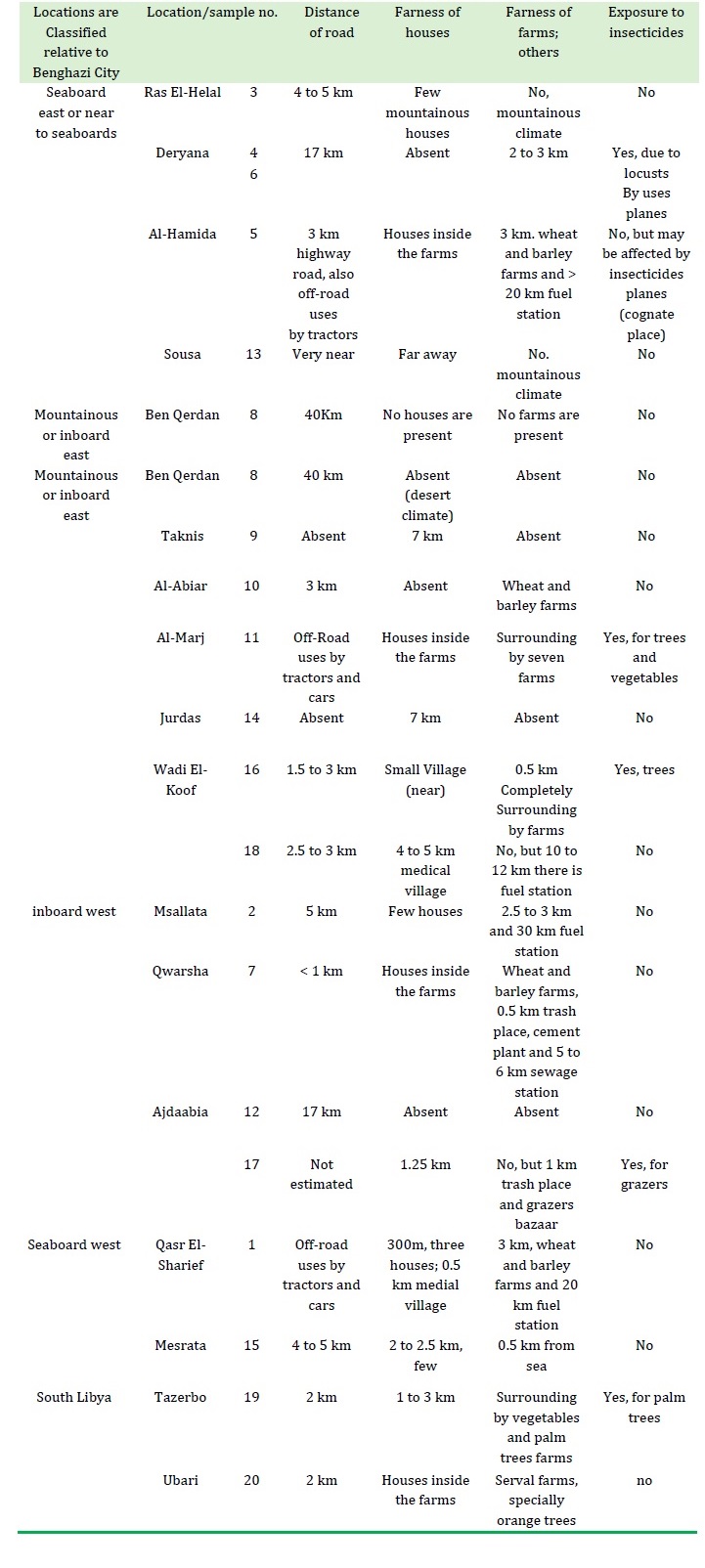
Table 2. The Location and type or name of honey samples
Results and Discussion
The results of this work are summarized in the following: The mineral content of honeys can be influenced by various factors, including temperature, humidity, soil, floral type, and other parameters [8]. Therefore, it is not possible to make definitive conclusions about the mineral content of honey.
This discussion will focus on nickel, cadmium, and lead, which are trace elements that have toxic properties [9]. If recommended amounts are exceeded significantly, other elements can also cause harmful effects [10].
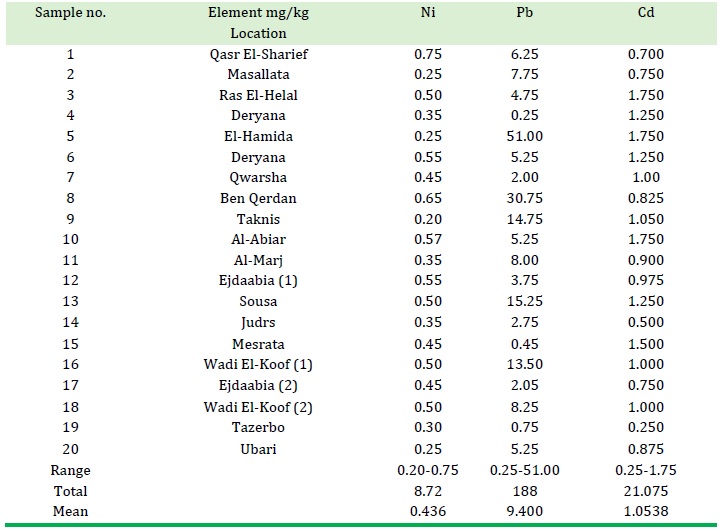
One of the elements that has been measured in honey samples is nickel. The concentration of nickel in the samples ranges from 0.20-0.75 mg/kg with an average of 0.436 mg/kg (as presented in the table). These findings are consistent with Romanian honey samples, which have a concentration range of 0.9 to 2.5 mg/kg [11].
Our results show lower ranges as compared to the honey samples obtained from Siena (Italy), where the range varies from 0.017 to 0.049 mg/kg [12].
The second component in our investigation is cadmium (Cd) which is a weighty metal that is toxic [8] and not necessary for human health [13]. Cadmium concentration in the honey sample ranges (0.25-1.75) mg/kg with a mean of 1.0538 mg/kg, as listed in Table 3. Therefore when we comprise our results with the Polish standard permits of Cadmium in honey which are (0.1 mg/kg) [14] and the maximum residues limits (MRL), proposed for the ER also (0.1 mg/kg) [15], we see all our results are higher than these limits.
We observed that all the Libyan honey samples analyzed in our study were found to be contaminated with a hazardous element.
Table 3. The concentration of elements (Ni, Pb, Cd) in different locations samples
As we previously mentioned, the source of this metal could potentially be from the steel or galvanized containers utilized during processing, shipment, or storage [16]. Industry, miming automobile exhaust gases may cause Cadmium contamination in honey [17,18].
Lead (Pb) is one of the three most significant toxic heavy metals [19].
The concentration of lead and cadmium in honey samples ranges from 0.25-51.00 mg / kg, with an average of 9.400 mg / kg, as provided in Table 3. All results indicate that each sample contains a certain amount of lead, and most of these samples have very high concentrations of this element. This high concentration implies that the honey samples have been exposed to lead pollution due to one or more of the reasons mentioned earlier. When we compare our findings with the approved limit of lead in honey according to the Polish standards, which is 0.4 mg/kg [15]. Furthermore, the EU has proposed a maximum residue limit (MRL) of 1.0 mg/kg [12].
All of our results exceed the specified limits, except for samples with numbers 4, 6, 15, and 19. However, the Czech bylaw 298/1997 permits a tolerable amount of lead up to 8.0 mg/kg. In comparison, the Romanian honey samples had a lead concentration ranging from 1.0 × 10-4 to 0.2 mg/kg [20].
The range for honey produced in England is between 0.002 to 0.2 mg/kg [21]. Honey in Siena (Italy) with a range from 0.0032 to 0.186 mg/kg [12], honey from Burgos (Spain) with range from 0.004 to 0.127 mg/kg [22].
Chilean honey, which ranges from 0.01 to 0.11 mg/kg [23], and Middle Anatolia honey (Turkey) which ranges from 0.0176 to 0.032 mg/kg [10], was much lower than our results.
Conclusion
All honey samples contain varying levels of certain elements, making it difficult to draw definitive conclusions about their origins. This is because honey, being a product of bees, can provide evidence of environmental pollution within an area of approximately 7 km2. Therefore, we collected some information from beekeepers about each sample under study to set some probabilities of the presence of these elements (Table 1). The levels of trace elements nickel, cadmium, and lead (Ni, Cd, and Pb) were measured and unfortunately, it was discovered that different levels of these elements are present in all honey samples analyzed. In other words, the honey has been contaminated with these trace elements, and we have outlined the likelihood of their presence. The level of nickel concentration in the sample was relatively low, and fortunately, it falls within the acceptable limits of nickel in food. However, it is believed that the main source of nickel presence is due to the use of steel or galvanized containers in different stages of processing, shipping, or storage. Another possible factor could be water supply used by the honeybees in their colonies, which may have been transported in galvanized containers. The elevated average lead concentration found in Libyan honey samples suggests that these samples have been exposed to lead pollution. However, the reliable interpretation of these results might be hindered due to those variables that are currently unknown. There is a possibility that some of the honey samples contain high levels of lead, possibly due to various factors such as insecticides, proximity to high traffic roads, human activity, or even the use of paint containing lead in the beehives.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Orcid
Najwa H Ansir
https://orcid.org/0000-0002-6998-3039
How to cite this manuscript: Najwa H Ansir*, khalid M darwish, Abdalslam Azzouz, Nuha El- Naas, Mohammed Y Gargoghil. Study and Estimation of Some Trace Elements (Ni, Cd, Pb) Content in Libyan Honey. Asian Journal of Green Chemistry, 7(4) 2023, 250-257. DOI: 10.22034/ajgc.2023.397344.1391


