Document Type : Original Research Article
Authors
- Alaa A. Omran 1
- Ola Hamad Salah 2
- Haider Falih Shamikh Al-Saedi 3
- Manal Morad Karim 4
- Hussam Abdali Abdulridui 5
- Anaheed Hussein Kareem 6
1 Department of Medical Engineering, AL-Nisour University College, Baghdad, Iraq
2 Department of Medical Engineering, Al-Manara College for Medical Sciences, Maysan, Iraq
3 Faculty of Pharmacy, Department of Pharmaceutics, University of Al-Ameed, Iraq
4 College of Pharmacy, National University of Science and Technology, Dhi Qar, Iraq
5 Department of Medical Engineering, Al-Hadi University College, Baghdad,10011, Iraq
6 College of Health and Medical Technology, Al-Ayen University, Thi-Qar, 64001, Iraq
Abstract
In this work, a coal fly ash (CFA) as a waste generated from chimney furnaces was tested as a low-cost adsorbent to streptomycin (SPM) drug removal from aqueous solution. Treatment of the samples coal fly ash was performed to reduce cost the of end use. CFA composition depends on the kind of coal utilized and has crystalline and no crystalline character. CFA is a valuable material and extensively utilized in cement production and as a higher adsorbent for water treatment. The physical properties like surface area, morphology, porosity, and chemical composition (alumina, iron oxide, silica, and titania) make CFA efficient material for wastewater treatment. The CFA was characterized via chemical and physical techniques, like FE-SEM, TEM, and EDX. The best optimum condition of adsorption method for SPM drug removal onto CFA, several factors were studied like, effect of contact time solution pH, concentration of drug, adsorbent dosage, and solution temperature. The percentage removal of SPM drug increased while the modified CFA dosage increased. The removal percentage % of drug increased with decreased drug concentration, also increased with increase quantity of CFA. The best of SPM drug removal found 91.76 % at concentration of drug 10 g/mL, adsorbent dosage 0.05 g, temperature 25 oC, and solution pH of 6.6. The adsorption models were tested with two isotherms like isotherm Langmuir, and isotherm Freundlich, the adsorption model was found to follow the model Freundlich.
Graphical Abstract
Keywords
Main Subjects
Introduction
In last years, the most world has turned to greater use of energy, so that energy production depends primarily on the health of living organisms and the environment. One of these problems is the massive consumption of coal, which produces very large quantities of coal fly ash (CFA), which is considered as the solid waste resulting from thermal power plants that rely mainly on coal. Therefore, there are environmental economic reasons to support the valorization of fly ash by reducing economic costs and eliminating solid waste landfills, and the main reason is the financial aspect that depends on sales of fly ash or products produced from the waste. Fly ash, a by-product resulting from the combustion of coal in thermal power plants, is an extremely prevalent and intricate anthropogenic substance. When not appropriately managed, it can lead to the contamination of water and soil, disrupt ecological processes, and present environmental risks. In recent times, there has been a notable increase in efforts to actively recycle fly ash [1-4]. For instance, approximately 20% of the fly ash produced is utilized in the concrete production. It is also employed in road base construction, soil amendment, zeolite synthesis, and serves as a filler in polymers, among other applications [5]. Nevertheless, the current applications of fly ash are not enough to fully utilize the generated amount. The leftover ash is considered waste and must be disposed of in ash ponds, lagoons, or landfills. However, stricter regulations for disposal sites, decreasing landfill space, and rising disposal expenses are expected in the future. Therefore, it is crucial to establish innovative recycling methods for coal fly ash. Ahmaruzzaman's (2010) review explored the characteristics and potential uses of coal fly ash [6]. In addition, there are several other studies that examine the various uses of fly ash, including soil improvement and mine restoration (Ram and Masto, 2010, Skousen et al., 2012, Ram and Masto, 2014) [7-9], extraction of alumina and cenosphere (Kolay and Bhusal, 2014, Yao et al., 2014) [10, 11], application as adsorbents for mercury removal, carbon dioxide capture, and wastewater treatment (Zheng et al., 2012, Wee, 2013), among other applications [12, 13]. Nowadays, several technologies are obtainable for the removal of pollutants from waste waters, like adsorption, biochemical ways, membrane separation, ion exchange, ultrafiltration, biological treatment, flocculation, precipitation, reverse, electro chemical processes, coagulation/flocculation, osmosis, photo degradation membrane filtration, precipitation, electrolysis, extraction mineralization, son lysis, advanced chemical oxidation, chemical reduction, and other processes. In recent years, several scientists are interested in the synthesis of the novel adsorbents for eliminating the organic and inorganic contaminants from waste waters via the adsorption process [14].
Adsorption has some advantage when compared aforementioned conventional techniques in terms the simplicity of use, effectiveness, low cost, etc. Though, several adsorbents have been investigated for the adsorption method of several kinds of contaminants from water and waste water, like chitosan, zeolite, silica, fly ash, coal, peanut hull, clay, natural phosphate, activated carbon, calcine alunite, and rice husk [15-21].
Streptomycin drug is an antibiotic medication and first discovered aminoglycoside, isolated from Streptomyces bacteria to utilized treatment a number of bacterial infections, like Mycobacterium valium complex, pulmonary tuberculosis, plague, brucellosis, tularemia, endocarditis, Burkholderia infection, and mouse bite fever [22]. Chemical formula C21H39N7O12, molar mass 581.580 g·mol−1, and chemical stretcher as show in Scheme 1.
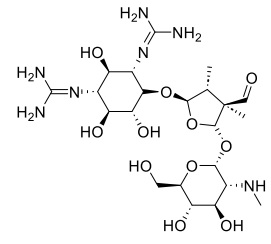
Scheme 1. Chemical structure of streptomycin drug
Experimental
Materials and methods
The materials used here were prepared from the Merck or Fluka (Switzerland) Company.
Preparation of fly ash
A quantity of fly ash used in this research is collected from bakery chimney furnaces in Hillah, Iraq. The fly ash is sieved through sieve to get rid of unwanted materials. First, fly ash is washed with water several times, then dried in an oven at a temperature of 65 oC for 5 h. Secondly, fly ash is activated by dousing in acid HNO3 (0.01 N) and stirring for 24 h, dried, and then burned at a temperature of 105 oC for 2 h. Black powder is obtained, placed in glass bottles and used in all experiments.
Adsorption experiments
Effect of weight of coal fly ash
The influence of CFA mass on the quantity adsorbed was found via agitating 100 mL of streptomycin drug solution of 10 mg/L in conical flasks 100 mL with adsorbent dosage of CFA (0.008-0.08 g) at 25 oC for contact time 1 h.
Effect of streptomycin drug concentration
100 mL of solution SPM drug in conical flasks 100 mL was prepared in several concentration (1-10 mg/L) and adsorbent of CFA 0.05 g at temperature 25 oC for contact time 1 h.
Effect of solution temperature
The influence of solution temperature on the quantity adsorbed was found via agitating 100 mL of streptomycin drug solution of 10 mg/L in conical flasks 100 mL with 0.05 g CFA at temperature (10-40 ᵒC) for contact time 1 h.
Effect of pH
100 mL of solution Streptomycin drug was prepared in a conical flask 100 mL with concentration of drug 10 mg/L and adsorbent dosage of CFA 0.05 g in conical flasks 100 mL and solution pH 2-10. The solution pH of drug solutions was adjusted with HCl or NaOH (0.01N) using UV-Vis spectrophotometer. The percentage removal and adsorption capacity of drug was determined utilizing Equations (1) and (2).
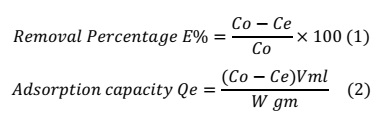
Results and Discussion
Characterization
FT-IR spectroscopy was conducted to identify the different functional groups present in the original fly ash (Figure 1). Symmetric stretching vibrations of Si-O-Si are detected at approximately 791 cm−1 while O-Si-O bending vibrations can be observed at around 475 cm−1. The presence of asymmetric Si-O-Al and Si-O-Si stretching frequencies can be determined by the absorption bands located at 1075 cm−1 and 551 cm−1, respectively. In addition, the existence of hydroxyl groups of silanols (–Si–OH) is indicated by a wide absorption peak observed at 3451 cm−1.
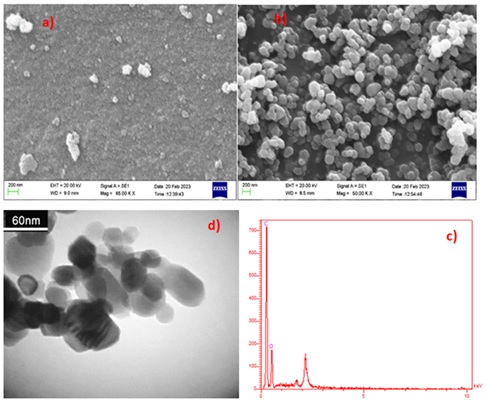
The surface morphology was studied by FE-SEM micrographs (Figure 2). As Figure 2a shows the FE-SEM microscopic images of the CFA samples, based on the FE-SEM analysis, it appears that most of the activated carbon has a heterogeneous, quasi-spherical morphology due to the occurrence of large aggregates in random shapes. Based on the results, it was confirmed that the use of activation produced pure CFA in less agglomerated and more homogeneous spherical shapes, which means that the acid activation and heat treatment preserved the regular spherical shapes of the carbon particles. In addition, in Figure 2b the FE-SEM micrographs of CFA after adsorption show its porous nature resulting from the filling of all surface active sites after the adsorption process.
The TEM images are as shown in Figure 3. The activated carbon had quasi-spherical shapes where small and almost dispersed spheres were obtained. Therefore, the large number of white spots between the disturbed carbon layers indicates that the activated carbon has porous structures [23,24].
The composition and purity of the prepared material were confirmed by EDX spectrometry and are demonstrated in Figure 4. The results reveal that the peaks are for C and O samples. EDX analysis indicated that the desired phases of O and C are present in the CFA with good purity.
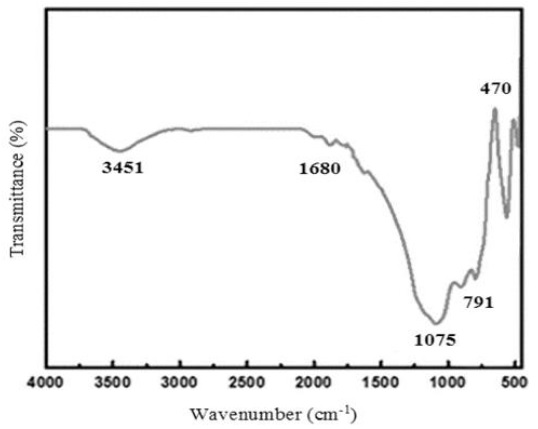
Figure 1. FT-IR of CFA

Figure 2. The FE-SEM image of a) CFA, and b) CFA after adsorption

Figure 3. The TEM image of the CFA
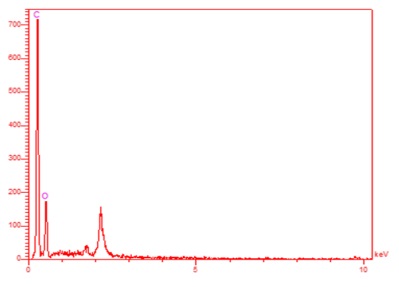
Figure 4. EDX analysis of the CFA
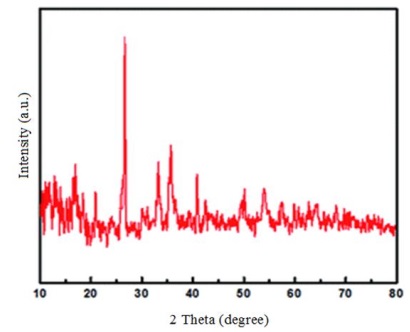
Figure 5. The XRD patterns of the CFA
X-ray diffraction (XRD) patterns confirmed the presence of quartz, mullite, and hematite phases in the original fly ash (Figure 5) [25]. The peaks are in good agreement with hexagonal quartz (JCPDS card no. 85-1054), orthorhombic mullite (JCPDS card no. 88-0107), and hematite (JCPDS card no. 88-2359).
Effect of pH solution onto adsorption SPM drug
pH solution plays significant role in adsorption method through influence on the target compounds, the charge density and species on the absorbance surface. In this research, the influence of solution pH is studied at range pH 2-10. The removal percentage % of SPM drug from aqueous solution via CFA is greatly dependent pH solution. The best condition pH solution at 6. The removal percentage % was the lowest at solution pH 2 (3.84 mg/g) and rising equal to pH 6, reached that the best (18.52 mg/g), as illustrated in Figure 6. At greater solution pH, the surface may get positively charged, which improves the negatively charged SPM anion through electrostatic forces of attraction [26].
Effect weight of coal fly ash
Influence of weight of CFA was needed in arranging to the minutest possible amount, which perform the best adsorption stoichiometric. The different amount of CFA from 0.01-0.15 gm /100 mL. The data appeared in Figure 7 show the removal percentage % of SPM against the weight of adsorbent CFA. Based on Figure 7, it is observed that the removal percentage % of adsorption is rises with increasing in weight of adsorbent CFA. This might be reflected to surface area rise of weight of adsorbent CFA, that rises the binding sites [27].
Effect of SPM drug concentration
The plot of the amounts of SPM drug adsorbed adsorption capacity (Qe) and removal percentage % SPM drug removal versus concentration of drug at different experimental conditions (Figure 8). The removal percentage decrease from (89.11%-97.99%) when concentration of SPM drug increase from (1-10 mg/L), while the adsorption efficiency (Qe mg/g) rise from (1.911 mg/g -19.344 mg/g) with increasing concentration of SPM drug. The elimination of SPM via adsorption on CFA was found to be fast low SPM drug concentrations, and then to slow down with rising in concentration drug. The elimination of SPM solution on to CFA via adsorption rose quickly at the start and then slowly slowed down until equilibrium was reached. It might be explained that a large number of vacant surface sites were available for adsorption throughout the initial stage [28,29].
The effect contacts time and solution of temperature
The contact time was measured by using the best conditions of adsorbent dosage, pH, temperature, and drug concentration. The aim is due to the occupation of the effective sites via the adsorbent dosage at the start, after which the rise is gradual until getting the contact time, as we notice that after this time there is no change in the quantity of adsorption as the absorbance is almost constant [30]. The effect of time and the amount of adsorbent for CFA was 19.344 mg/g, as shown in Figure 9a.
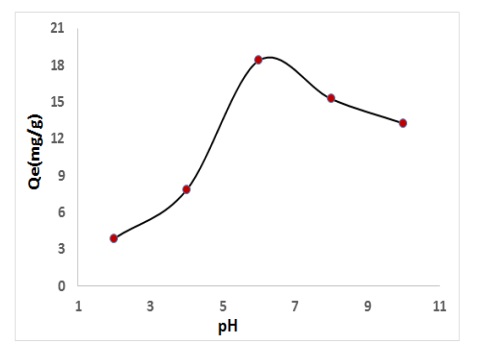
Figure 6. Effect of pH solution on to removal percentage of drug using CFA
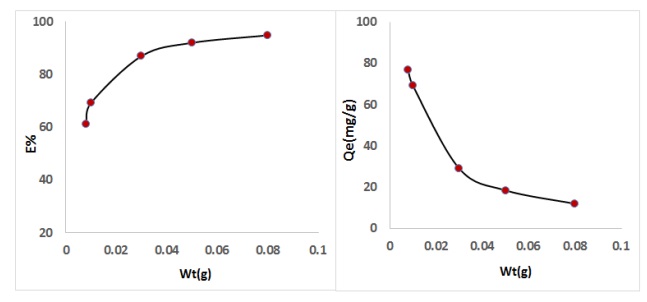
Figure 7. Effect of weight of CFA on to removal percentage of drug
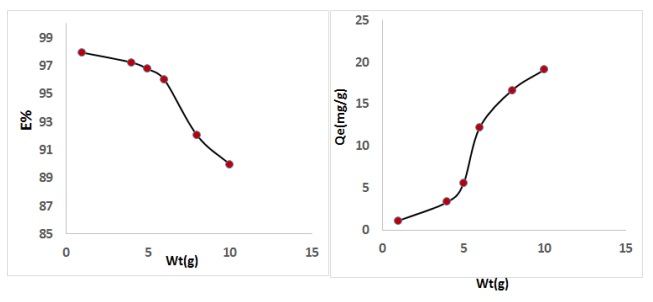
Figure 8. Effect of SPM drug concentration
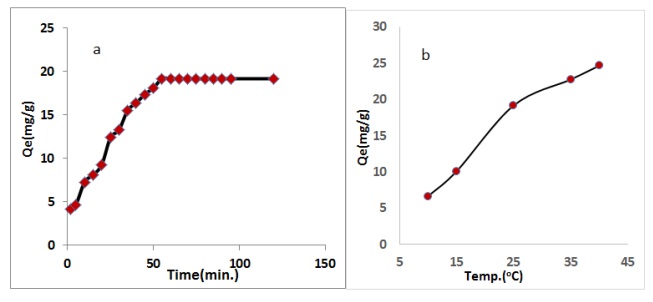
Figure 9. Effect of a) contact time and b) solution of temperature onto adsorption capacity of drug by use CFA
A significant parameter of the adsorption method depends on the solution temperature. The influences of solution temperature were carried out at several temperatures (10-40 oC). The adsorption capacity Qe mg/g increase of adsorbate with increased temperature solution. Adsorbent surfaces that no affected via the molecules' adsorption, which is why increasing the solution temperature may lead to the break-down of some active functional groups on the rim surface composite, cause in a reduction in the number of active sites, as shown in Figure 9b [31, 32].
Adsorption isotherm
Isotherms adsorption models can provide some significant result about the distribution of adsorption molecules among the liquid and solid phases, and can provide vital data about the interactions among adsorbent and adsorbent at a given concentration. In this study, the data from the influence of drug concentration was adopted to estimate the nature of removal via using adsorption isotherm, model Langmuir, and model Freundlich, as presented in Table 1, as model non-linear curves of adsorption models of drug on to fly ash [33]. The equilibrium mechanisms its well to isotherm Freundlich model for removal drug, as listed in Table 1. Obviously, the Freundlich model isotherm explain the spreading of drug among the liquid states and solid states on the basis of prospects associated upon the varied or unvaried situations of the fly ash. The equations of two isotherm Langmuir and Freundlich nonlinear was calculate in Equations (3) and (4), respectively [34, 35].

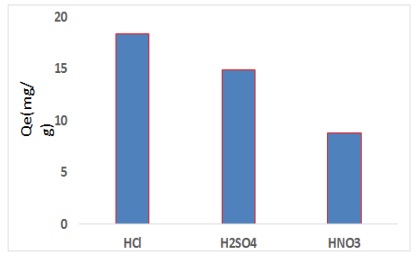
Figure 10. Influence of acid treatment on CFA surfaces
Influence of acid treatment on surfaces CFA
The study influence of acid treatment was essential to illustration the best adsorption and removal percentage. The CFA adsorbent was treated via several acids like (H2SO4, HNO3, and HCl). The data appear in Figure 10. It was obvious from the data appear in Figure 10, the top adsorption efficiency when the adsorbent treated via HCl, this is may be due to the raised in acid acidity caused to re-activated the active sites for CFA surface.
Conclusion
In this investigated, preparation of low-cost adsorbent from coal fly ash and removal of streptomycin drug from wastewater. The adsorption efficiency of drug increased with increasing weight of CFA and temperature, but concentration of drug decreases with adsorption efficiency increases for constant weight of CFA. adsorption efficiency (Qe mg/g) rise from 1.911 mg/g-19.344 mg/g with increasing concentration of SPM drug thus in case of drug higher value of pH, increased adsorption efficiency increases. The removal percentage % was lowest at solution pH 2 (3.84 mg/g) and rising equal to pH 6, reached that a best (18.52 mg/g). Also, the equilibrium isotherm was well described via Freundlich isotherm.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the article and agreed to be responsible for all the aspects of this work.
Orcid
Alaa A. Omran
https://orcid.org/0000-0002-5688-447X
Ola Hamad Salah
https://orcid.org/0000-0002-3194-1749
Haider Falih Shamikh Al-Saedi
https://orcid.org/0000-0003-0862-9543
Anaheed Hussein Kareem
https://orcid.org/0009-0008-8881-6122
How to cite this manuscript: Alaa A. Omran*, Ola Hamad Salah, Haider Falih Shamikh Al-Saedi, Manal Morad Karim, Hussam Abdali Abdulridui, Anaheed Hussein Kareem. Evaluation of Activity Coal Fly Ash as Low Cost an Adsorbent for the Removal of Pollutants: Equilibrium and Isotherm Studies of Pharmaceutical Compounds. Asian Journal of Green Chemistry, 8(2) 2024, 161-172. DOI: 10.48309/ajgc.2024.425385.1460