Document Type : Review Article
Author
Dr. Akhavi Laboratory Co, Scientific Department ,Tehran, Iran
Abstract
This article is the first comprehensive research that review of the potential benefits of Punica granatum in dermatology. Currently, since plant extracts are used in skin, hair, and cosmetic industry far more than before, concerning that pomegranate is native to Iran, this overview of its properties in skin diseases field is based on scientific research of this valuable fruit.
Graphical Abstract
Keywords
Main Subjects
Introduction
Knowledge about pharmaceutical herbs add novel horizons to the world of medicine. Herbal medicines are cost-benefit and safer in comparison to synthetic medicines. Much of the data is limited to traditional healers or passed to the next generation orally, so it is important to preserve this valuable knowledge before it disappears [1].
Since ancient times, pomegranate (Anar in persian) has been employed to remedy many diseases [2, 3]. Cultivation and edible use of Punica granatum may date back to 3000 BC. Moreover, different portions of pomegranate have been utilized in traditional medicine to remedy different diseases. Pomegranate fruits to repel parasites, seeds and fruit peels to treat diarrhea, flowers to manage diabetes, bark, and roots to stop bleeding and repairing wounds. Likewsie, the leaves are used to control of inflammation and treatment of gastrointestinal disorders [4].
P.granatum L. belongs to the Punicaceae family (Figure 1) [5] and is native shrubs in central Asia, specifically some districts of Iran. Pomegranate has many potential health benefits and it has clinical trials in different diseases. Its scientific name is Punica granatum, which is usually called pomegranate, is mainly found in Iran, which is the main center of this valuable fruit with therapeutic properties [6].
According to the agriculture ministry, Iran is the world’s 3rd biggest producer with a production of 1.1 million tons per year and a cultivated area of around 90,000 ha [7]. P. granatum is a tree about 5 to 8 meters that is mostly localized and originated from Iran, it is also found in Afghanistan, Pakistan, India, PRC, the United States of America, and all over the Mediterranean region [8].
In ethnobotanical study on the therapeutic use of P. granatum, it was found that this herb has a variety uses for a broad range of common diseases such as scabies and cholera. A part of the whole plant might be use alone or in combination with other plant or minerals to improve its effectiveness [9].
Pomegranate has numerous medicinal activities, such as anti-diabetic, anti-inflammatory, anti-malarial, anti-fungal, and anti-bacterial. It also has effects such as improving the gasterointestinal tract microbiota, preventing obesity, and improving fertility in men. Pomegranate is further used to treat some diseases, such as cardiovascular problems, cancer, depression, and wound healing [10].
Fruit peel powder is used to accelerate wound healing, as a purgative, and to relieve indigestion. Astringents, laxatives, and blood purifiers are all found in pomegranate fruit. In traditional medicine, leaves, fruits, fruit skin, and seeds are commonly used for various diseases.
Fruit skin (32%) was the most used in medicine production, followed by seeds (22%) and flowers (20%), whole plant (9%), shoots (7%), root bark (6%). %, seed coat and root (6%), and whole plant (9%) (each 2%) (Figure 4) [11]. P. granatum species are major native herbs to Iran, which grow in arid and semi-arid areas because of their potency to adjust to unfavorable environmental conditions in most areas of the country.
Pomegranate has many health benefits so that this heavenly fruit is mentioned three times in the Holy Quran [12]. P. granatum is broadly used to cure dysentery, bacterial infections, diarrhea, hemorrhage, and respiratory diseases [9].
The current research is the first comprehensive investigation about use of pomegranate on cosmetic and dermatological problems.
The studied databases included Springer, Hindawi, PubMed, Science Direct, Google scholar, and other scientific resources. The data acquired through the different valid sites in the current research has been compiled, interpreted, and presented using key words such as in this study, more attention was paid to the skin benefits of pomegranate in skin problems and unrelated articles were not considered. In this review article, 41 related research articles and their results were mentioned.

Figure 1. Pomegranate tree, flower, and fruit [3]
Phytochemistry
Pomegranate pericarp has a high amount of phytochemicals, mainly polyphenolic flavonoids, and ellagitannins, containing ellagic acid and punicalagin [13]. Pomegranate contains phytochemical compounds in its diverse parts, which have several pharmacological functions (Tables 1 and 2) [14].
Pomegranate juice
Anthocyanin creates an attractive purple-red color in pomegranate juice, which is reduced during the pressing process. The amounts of fatty acids, sterols, organic acids, triterpenoids, and minerals content have been also reported in pomegranate juice [20]. In general, pomegranate has two main composition groups:
Polyphenols
Hydrolysable tannins are the principal group of pomegranate polyphenols, which consist of gallotannins, ellagitannins, galagyl esters, hydroxycinnamic acids, and hydroxybenzoic acids.
The prime ingredient of ellagitannins is punicalagin, which is mostly recognized in the pericarp, peel, flowers and seeds. In addition, punicalagin and its isomers, pomegranate contains punicalin A, punicalin B, and pedunculagin isomers. Likewise, Gallic acid, ellagic acid, caffeic acid, chlorogenic acid, p-coumaric acid, aglycone, and ferulic acid are present in pomegranate.
Table 1. Phytochemicals of P.granatum [14]

Table 2. Pharmacological activities of the P. granatum extracts [27]
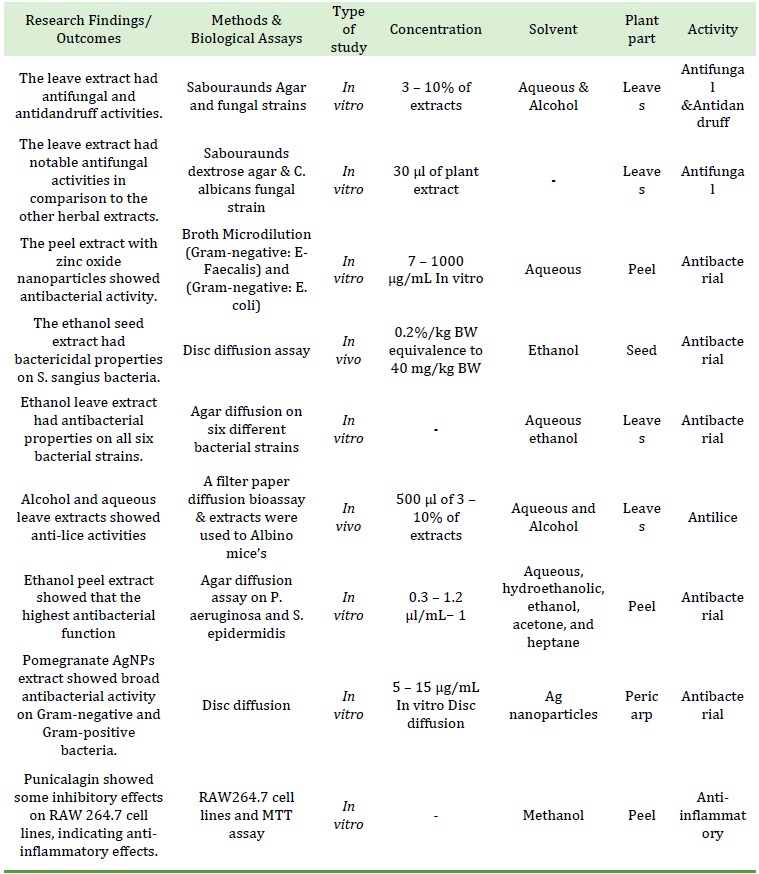
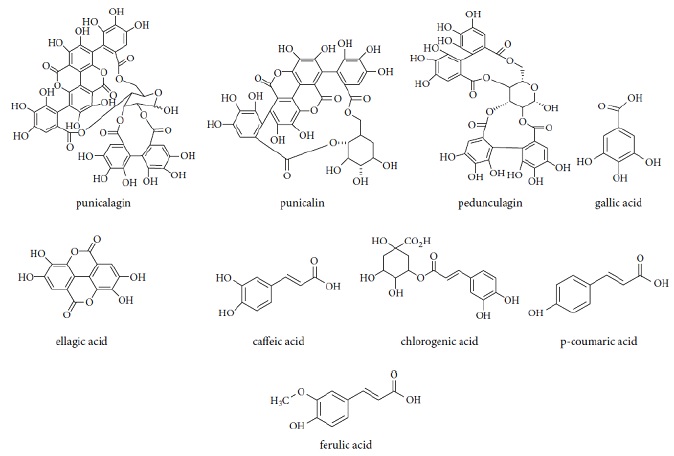
Figure 2. The important phenolic ingredients in P.granatum [21]
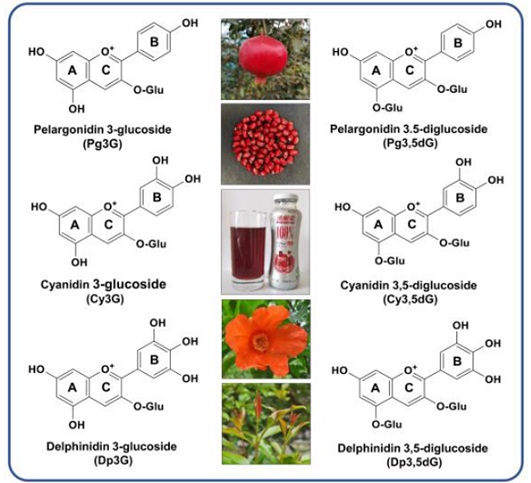
Figure 3. The commonly main Anthocyanosides ingredients in P.granatum [24]
Figure 2 displays the important polyphenol ingredient of pomegranate [21].
Anthocyanosides
They are the next significant ingredient group that exists in the flower, fruit, and are accountable for the red color of arils (Figure 3) [21, 22]. Pomegranate is rich in anthocyanin, which has significant antioxidant capacity. Anthocyanins mainly act as primary antioxidants [23].
Pomegranate seed oil (PSO)
PSO contains 30.12% punicic acid, 28.92% linoleic acid, 19.01%, oleic acid, 1.49% gadoleic acid, 0.18% erucic acid, 0.18% linolenic acid, and 0.07% palmitoleic acid (Figure 4) [25].
Pomegranate peels
Pomegranate skin has various forms of tannins, which are water-soluble and hydrolysable phenolics. According to the structure, tannins have four main categories: gallotannins, ellagitannins, complex tannins, and dense tannins. Punicalagin is an ellagitannin which is the major component in the peel of pomegranate, whose content is much higher than other compounds (Figure 2) [26].
- granatum in traditional medicine
In traditional medicine, pomegranate peel, flowers, branches, and roots are used. All P. granatum L. components have strong astringent effects due to the presence of abundant tannins. Several pomegranate flower infusions and decoctions of the flowers have been utilized to remedy diarrhea and vaginal infectious discharge. In addition, the extract of pomegranate peel has been used to treat acute pancreatitis.
Fresh pomegranate juice is use for remedy of gall bladder problems and its decoction is useful for any types of diarrheas, either simple or bloody diarrhea and stomach discomfort. Alkaloid substances in pomegranate root bark and ethanolic extract are used to eliminate intestinal parasites. Also, its antibacterial and anti-inflammatory effects was utilized in traditional medicine [22].
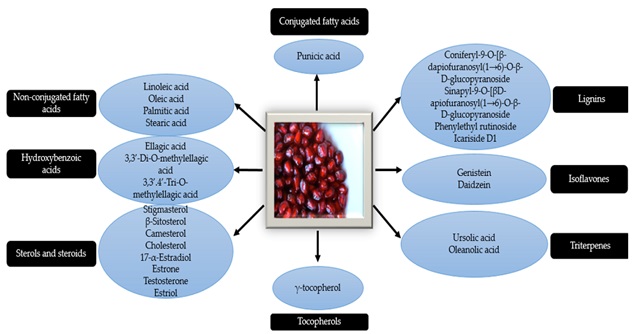
Figure 4. Chemical composition of the P.granatum seeds [21]
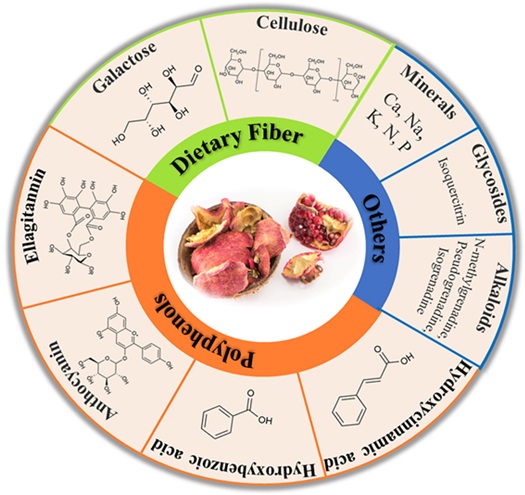
Figure 5. Chemical composition of P.granatum peels [26]
- granatum in dermatology
- granatum L. is a shrub that contains specific components in particular polyphenols and anthocyanins, which have antioxidant, antiinflammation, wound healing, whitening, antiacne, anti-hair loss, antiviral, and antibacterial effects. It also prevents skin photoaging due to UVB [22].
Wound healing activity
Several research have proven P. granatum improve wound repairing process. Nayak et al. assessed the wound repairing process of P. granatum fruit peel. In this study, a 95% decreasing wound area was reported on animals treated by the extract compared with 84% at control group.
Skin epithelialization in pomegranate extract-treated group was faster than control group, and the content of hydroxyproline in pomegranate extract-treated group was remarkably greater than the control group. As a result, P. granatum significantly improved wound healing in rats [23].
In addition, Murthy KN et al. discovered that pomegranate extract had high phenolic compounds (44%). They formulated a 10% water-soluble gel to investigate wound healing, skin contraction percentage, collagen, and hydroxyproline content in a rat skin model.
Rats treated with 2.5% gel showed a mean improvement of 55.8% and 40.8% compared to the positive and negative control groups, respectively. Whilst, with 5.0% gel group exhibited well repairing of 59.5% and 44.5% in comparison to the negative and positive controls, respectively.
Hydroxyproline content was enhanced in the 5% gel treatment. Histological evaluation confirmed wound repairing in the use of gels containing extracts. The content of hydroxyproline in the 5% gel increased twice as well as histopathological evaluation support wound healing with the use of gels. The wound healing was by 5.0% gel, 5.2% gel, and control, 10, 12, and 16 to 18 days, respectively. Chromatography results showed catechin and gallic acid as the main components of their repair [28].
In a study, ointments of methanolic extract of P. granatum at 10%, and 15% w/w were investigated for the effect of wound healing in a cut wound model of rats. The results were as same as those of nitrofurazone ointment. Wound contraction activities of 10% and 15% P. granatum ointments were 97.8%, and 98.4%, respectively, and significantly (P < 0.05) higher than the control. In the 15% w/w pomegranate extract ointment group, wound closure time and wound contraction percentage were lower and higher, respectively. In the 10% pomegranate extract ointment group, wound contraction was significantly observed on the 18th day and wound closure time attained 100% on the 20th day. Therefore, methanolic extract of pomegranate demonstrated as a potential wound healing agent [29].
Pirbaluti et al. used the diethyl ether extract of P. granatum and M. sylvestris flowers to investigate wound healing effects in a dose of 200 mg/kg/day in alloxan-induced diabetic rats. They divided rats into six groups, the groups treated with M. sylvestris and P. granatum compared to the control, and other groups, including the nitrofurazone group, effectively caused wound contraction, increased collagen, and increased fibroblasts. These findings indicated that these two herbal extracts have the ability to manage wounds [30].
These findings indicated that these two herbal extracts have the ability to manage wounds [30].
Nasiri et al. assessed the activity of pomegranate flower extract and silver sulfadiazine in the treatment of burn damage of rats. In this study, 10 Wistar rats were used in each group with topical cream base, normal saline, cream containing 1% silver sulfadiazine, and 5% and 10% whole pomegranate creams until the wound area is completely healed; the healing time of lesion contraction percentage and histopathologic characteristics including neovascularization and collagenization were examined. An interesting point to note was the reduction in wound size with cream containing pomegranate extract on day 15 in mice of this group compared to silver sulfadiazine, and on day 25 of the group treated with pomegranate extract, the wounds were completely healed. These results show that the extract of pomegranate might has improved wound healing and can be helpful in the cure of burn wounds [31].
Anti-inflammatory activity
Ellagic acid (EA) possess anti-inflammation effects in chronic disorders. Also, Ellagic acid treatment suppresses inflammation responses on keratinocytes by regulation vital inflammation signaling pathways like mitogen-activated protein kinases, signal transducers, and activators of transcription (STAT).
In vivo tests on a DfE-induced Atopic dermatitis mice exhibited that the EA improved skin signs by reducing inflammatory responses. Therefore, EA can be a potential alternative treatment for atopic dermatitis by inhibiting inflammatory signaling pathways [30].
Due to several risks related to NSAIDs for the remedy of pain and inflammation, medicinal plants are used as alternative therapies. Ben Saad et al. showed that ellagic acid, gallic acid, and punicalagin A&B potentially inhibited the production of NO, PGE-2, and IL-6 induced by LPS, and probably ellagic acid, gallic acid, and punicalagin which are the most important components of pomegranate and they are probably responsible for role anti-inflammatory [32].
Antibacterial and antimicrobial activity
Tannins may show their antimicrobial activity via three mechanisms: inhibition enzymes function, precipitation of membrane proteins, and reduction of metal ions [33].
Herpes disease is always in need of new and effective treatment products, Therefore, Houston et al. evaluated the activity of pomegranate fruit and zinc metal ions on herpes simplex. In this study, the effect of this compound on herpes simplex type 1 and acyclovir-resistant herpes in host cells of Vero was done by MTS method and commercial kit. Zinc sulfate, zinc citrate, zinc stearate, and zinc gluconate indicated increased antiviral activity similar to PRE against HSV-1 up to 4 times. Punicalagin had eight times more antiviral activity than an equivalent mass of pomegranate rind extract (PRE). Even so, the antiviral information displayed that punicalagin has viricidal activity remarkably lower than that of PRE, which is comparable to acyclovir.
Likewise, PRE exhibited its potential versus acyclovir-resistant HSV, whilst acyclovir exhibited no potency. The enhanced antiviral action of PRE in combination with Zn (II) has the potential as an innovative topical drug versus HSV infections like cold sores [33].
- granatum has known as an herbal drug with potential antimicrobial activity. Recently, Houston DM et al. has been indicated that the antiviral effect of pomegranate rind extract is striking increased with zinc (II) ions with a potency of up to 7 log decline versus herpes simplex virus (HSV). Vesicular eruptions of HSV1 and HSV2 lesions induce a significant inflammation in local area, leading to erythema, swelling, and pain, especially in the later stages of infections [33].
Sadeghian et al. evaluated pomegranate peel extract as an antibacterial compound against Gram-positive bacteria such as Staphylococcus aureus, Gram-negative Pseudomonas aeruginosa, and pathogenic yeasts like Candida albicans. The antibacterial activity of aqueous and methanolic extracts was compared with antibiotics using three different techniques: agar dilution, cylinder plate, and antibiogram.
The results indicated that extracts had significant bactericidal effect on Staphylococcus aureus and P. aeruginosa. In addition, the methanolic extract had a potent antifungal activity on C. albicans. Antimicrobial effects on Staphylococcus aureus, P. aeruginosa, and C. albicans were similar to cloxacillin, gentamicin, and clotrimazole, respectively. Methanolic pomegranate fruit peel extract was more effective than aqueous extract on all tested bacteria. Therefore, the P. granatum extracts can be a great source for new antibacterial drugs [34].
Duman et al. evaluated the arils of six P. granatum L. cultivars of Turkey in term their antimicrobial activity by agar diffusion and minimum inhibitory concentration (MIC) methods on seven bacteria: Bacillus megaterium, Pseudomonas aeruginosa, Staphylococcus aureus, Corynebacterium xerosis, Escherichia coli, Enterococcus faecalis, and Micrococcus as well as three fungi (Kluvyeromyces marxianus, Rhodotorula rubra, and Candida albicans).
Aril Pomegranate extract has an antimicrobial activity on all microorganisms and giving inhibition zones of 13 to 26 mm [35].
The MIC values for the active extract were between 30 and 90 µg/ml. The results seem to support the antimicrobial potential of P.granatum species [36].
Antioxidant activity
Polyphenol hydroxyl groups in pomegranate are responsible for antioxidant activity by scavenger activity free radicals. Likewise, the catechol hydroxyl groups of complex and condensed tannins have the potency to chelate iron and transition metals [13].
Pomegranate is a rich of anthocyanin, which has a potent antioxidant capacity. Gill et al. found that phenolic ingredients are responsible for 28% of the total antioxidant function of pomegranate, which indicates the important role of anthocyanin in its antioxidant [22].
There are several articles about the antioxidant activity of pomegranate, mostly based on the DPPH assay. In general, the parts of pomegranate have significant anti-radical and anti-microbial effects, which can make this plant introduced safe as an herbal source of preservatives and antioxidants. Therefore, P. granatum might be used as an auxiliary substance as mentioned above in pharmaceutical and food industries [35].
Skin whitening activity
Topical microemulsion containing P. granatum extract controls erythema and redness of skin as well as skin melanin in people. Since P. granatum is rich in polyphenolic compounds, it shows its function with strong activity in inhibiting free radicals. It is believed that ellagic acid acts as a skin brightener by chelating copper in tyrosinase. This research shows that the microemulsion of P. granatum extract could be utilized in a topical formulation for skin spots and erythema [37].
Today, there are many effective ingredients and skin-lightening products that may inhibit tyrosinase activity in the melanogenesis process. Tyrosinase inhibitors in particular hydroquinone, kojic acid, and arbutin may induce skin irritation or acute dermatitis, and there are many concerns about the safety of these agents. Thus, the need for safe and effective natural skin lightening agents has increased more than ever. Pomegranate fruit extract contains substances that inhibit melanogenesis and shows its potential to be used as a lightening cream in cosmetic formulations. punicalagin is a polyphenolic compound in pomegranate fruit extract, that has been recognized as a melanin production inhibitor [38].
New studies show that the extract of Larix sibirica and P. granatum in a ratio (1:1) caused a two-fold decrease the melanin quantity in the cells that the larch extract or Siberian pomegranate alone could create [38].
Anti-acne activity
Acne vulgaris is a prevalent dermatology disease associated by excessive sebum production, hyperkeratosis, and the presence of Cutibacterium acnes, Staphylococcus aureus, and inflammation. Lee et al. indicated that pomegranate extract remarkably decreased swelling by C. acnes of the Wistar rats' ears [39].
Their results represented that pomegranate extract inhibited bacterial growth and lipase function. Four hydrolysable tannins are punicalagin, punicalin, strytinin A, and also granatin B. Punicalagin and punicalin compounds had antibacterial properties and testosterone-induced HaCaT proliferative effects more than others. punicalagin, strictinin, and granatin B showed lipase inhibitory effects. Granatin B compound causes downregulation of cyclooxygenase-2 expression and prostaglandin E2 production in RAW 246.7 cells treated with P. acnes.
As a result, pomegranate hydrolysable tannins show strong anti-acne, anti-lipase, anti-keratinocyte proliferation, and anti-inflammatory activities. Therefore, pomegranate extract (PG-E) has major potential for topical use in anti-acne agents, and punicalagin appears to be the most effective pomegranate compound and quality control indicator [32].
Anti-skin aging activity
The Ultraviolet (UV) radiation causes various skin problems, including sunburn, inflammation, hyperplasia, immune system suppression, skin aging, and skin cancer. Using the GC-MS method, Park et al. showed that pomegranate polyphenols help to prevent and protect the adverse skin effects of ultraviolet rays. To consider the protective pomegranate activity of skin aging caused by UVB, they determined the level of procollagen type I and MMP-1. Finally, the dominant compounds were catechin, quercetin, kaempferol, and equol. Sunlight induces changes in the expression of procollagen type I and MMP-1 in fibroblasts. Studies showed that pomegranate catechins, which are one of its main polyphenol compounds, can play a main role in protecting against skin damage caused by UVB [40].
Many in vivo and in vitro tests have been indicated that polyphenols have antioxidant, anti-inflammation, and anti-cancer activity. It seems that pomegranate fruit extract standardized to punicalagins had effective in keep fibroblasts apoptosis after UV radiation, probably by reducing the activation of pro-inflammation transcription factor NF-kappa B a downregulation of proapoptotic caspase-3, and an increased G0/G1 phase, linked to DNA repair. The results of this survey illustrated the protecting properties of pomegranate extract versus UVA and UVB-induced apoptosis and the possible application of extract polyphenols for topical formulas [41].
The pomegranate fruit extract has a potent antioxidant and anti-inflammation hallmark. The current research has indicated that pomegranate fruit extract treatment of human epidermal keratinocytes prevents UVB-mediated activation of MAPK and NF-kappa B pathways. Signal transducers and activators of transcription 3 (STAT3), Protein Kinase B/AKT, and Map Kinases (MAPKs) regulate cell proliferation, apoptosis, and other biologic pathways. Pomegranate protects skin against UVA-mediated activation of STAT3, AKT, and extracellular signal-regulated kinase (ERK1/2). Our data indicated that PG is an effective agent for ameliorating UVA-induced damage and it is worth further evaluation as a photo preventive agent [42].
Anti-stretch marks
Stretch Marks called as dermal lesions with multifactorial mechanisms had not been precisely explicited so far. However, striae scarcely cause serious medical issues, they present a cosmetic concern for sufferers. Bogdan C. et al.'s study on the clinical effectiveness of a PSO extract and C.lechleri cream formulation indicated that it could be useful in preventing or improving striae [43].
Hair growth promoting activity, anti-dandruff and cure of head lice
It seems that use of Punica granatum in skin and hair diseases as well as cosmetic industry may be beneficial. According to a new research, pomegranate extract may be useful for prevention of hair loss, as anti-dandruff and cure of head lice. New research is need for potential use for other application of this precious local plant [44].
Hair color protection
Liposomal pomegranate peel extract formulations may protect hair color due to the antioxidant properties and the film-forming activity of the liposome formulation. As a result, the liposomal formulation may be a suitable alternative to reduce the dyeing of frequency and protect hair fibers [45].
Sunlight fades both natural and dyed hair by inducing free radicals, which oxidize hair melanin, and antioxidants can reduce these color changes. Pomegranate has a high amount of polyphenol compounds and tannins, which have very high antioxidant properties.
In a study, formulas containing silicon and combined formulas of silicon and pomegranate were compared in stabilizing hair color. 5.0% and 10.0% w/w pomegranate extract were found to be effective in preventing hair color fading by 37.6% and 60.8%, respectively.
Silicone creates a protective layer on the hair fiber, while pomegranate extract can protect hair color by preventing oxidation.
However, the simultaneous use of extract and silicone in the formulation produced controversial results [10].
Conclusion
Punica granatum L (pomegranate) which contains specific ingredients like polyphenols and anthocyanins, possess potential antioxidant, anti-inflammatory, wound healing, whitening, anti-acne, and antibacterial effects. It is also viricidal against herpes simplex virus infection and prevents skin photoaging due to UVB.
Acknowledgments
I would like to thank my dear wife, Monireh Talajoran, for her invaluable guidance and support throughout the research process.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Orcid
Farshad Akbarnejad
https://orcid.org/0000-0001-9774-0985
How to cite this manuscript: Farshad Akbarnejad *. Dermatology Benefits of Punica granatum: A Review of the Potential Benefits of Punica granatum in Skin Disorders. Asian Journal of Green Chemistry, 7(3) 2023, 208-222. DOI: 10.22034/ajgc.2023.388077.1388


