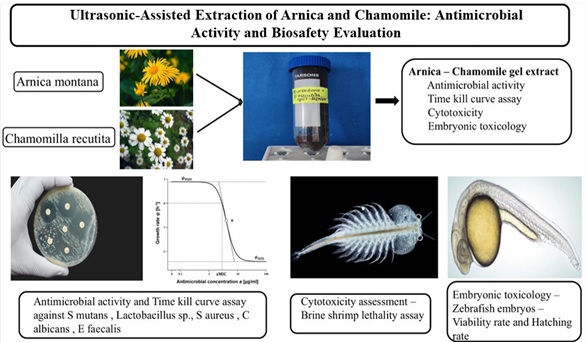
Ultrasonic Extraction of Arnica–Chamomile Gel: Antimicrobial and Safety Evaluation
Pages 181-195
https://doi.org/10.48309/AJGC.2026.542761.1816
Sharuniveda Sankaranarayanan, Umamaheswari Thirupamburam Natarajasundaram
Abstract Herbal formulations are gaining attention as complementary options to conventional therapies due to their phytochemical diversity, favorable safety, and broad therapeutic potential. Arnica montana and Chamomilla recutita are particularly recognized for antimicrobial, antioxidant, and anti-inflammatory properties, making them suitable for novel biomedical applications. In this study, an Arnica–Chamomile gel was prepared using aqueous extracts and evaluated for antimicrobial efficacy and preliminary safety. The formulation was tested against bacterial and fungal strains using standard assays, with time-kill kinetics providing insight into dose- and time-dependent activity. Safety was assessed through brine shrimp lethality and zebrafish embryotoxicity models. Results indicated promising antimicrobial effects, especially against bacterial strains, with acceptable tolerability at therapeutic levels. However, higher concentrations showed variability, underscoring the need for optimization before clinical translation. These findings support the potential of Arnica–Chamomile gel as a sustainable, plant-based preparation and highlight the relevance of traditional botanicals in modern biomedical research.