Benzimidazolium-Catalyzed Cross-Coupling of Aromatic Aldehydes and Benzyl Halides in Water: A Green and Efficient Synthesis of 3-Hydroxy-1,2,3-Triarylpropan-1-Ones
Pages 1-15
https://doi.org/10.48309/AJGC.2026.530868.1759
Baramee Phungpis, Pakin Noppawan, Kanokkan Worawut, Potchanee Keawkumsan
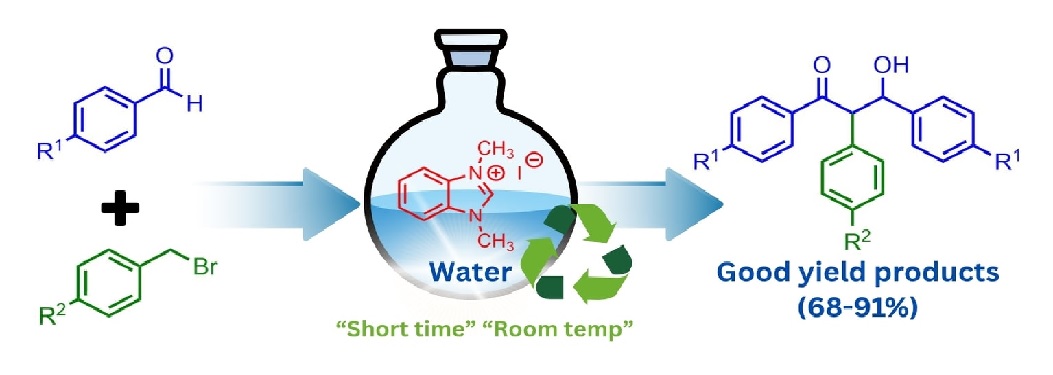
Abstract The development of green reaction conditions using H2O as the reaction medium enabled the efficient reaction of various aromatic aldehydes and benzyl halides. The reactions were carried out under mild conditions using 50 mol% N, N-dimethylbenzimidazolium iodide and 50 mol% NaOH, with reaction times optimized for each substrate. This protocol yielded 3-hydroxy-1,2,3-triarylpropan-1-ones as the main products, formed through the further reaction of initially generated α-aryl ketones with an additional equivalent of aldehyde under basic conditions. The desired products were obtained in good to excellent yields (68-91%). A minor amount of α-hydroxy carbonyl compounds was consistently observed as a by-product. Notably, the aqueous reaction medium containing the benzimidazolium salt and NaOH was recyclable and could be reused for at least three consecutive cycles without a significant decrease in catalytic activity.