Document Type : Original Research Article
Authors
Department of Chemistry, College of Sciences, Shiraz University, Shiraz 7194684795, Iran
Abstract
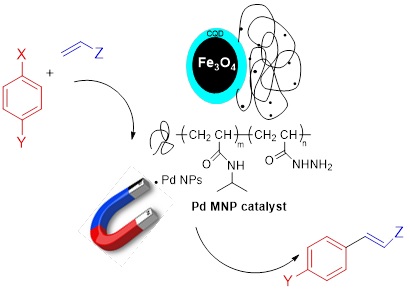
A new smart Pd catalyst was created based on functionalized thermo-responsive poly (N-isopropylacrylamide) (PNIPAM) grafted Fe3O4@CQD@Si. For catalyst fabrication, initially, Fe3O4 magnetic nanoparticle was prepared by co-precipitation method, and then it was coated with CQD as sustainable and environmentally friendly support. Chemical modification of Fe3O4@CQD support with polymerizable groups was performed through vinyltriethylsilan (VTES) reaction. Thereafter, PNIPAM was grafted to the modified Fe3O4 via the conventional free radical polymerization technique. The PNIPAM-grafted Fe3O4@CQD@Si was further treated using hydrazine to create the modified amino support, which was then complexed with Pd(OAc)2 to yield the magnetic heterogeneous catalytic system. All materials are characterized using various methods, i.e., FT-IR, SEM, DLS, CHN, XRD, zeta potential, VSM, and TGA analysis techniques. Fe3O4@CQD@Si@PNIPAM-NH2/Pd catalyst was used to produce a range of substituted alkenes in the Mizoroki-Heck cross-coupling reaction with a high turnover number (TON) and good to outstanding yields. Above the LCST temperature of the PNIPAM, the hydrophobic part of the polymer starts to shrink, and this smart catalyst advances the relevant reactions in milder conditions. Easy purification of the substituted alkenes due to the convenient isolation of the magnetic catalyst with an external magnet is the main characteristic of this process.
Graphical Abstract
Keywords
Main Subjects
Introduction
One of the noteworthy and broadly applied transformations for the creation of bonds between carbon atoms is the Heck reaction, allowing various alkenes to get vinylated, arylated, or alkylated mildly in a single step by reacting them with benzyl, vinyl, aryl, acetates, triflates, or allyl halides applying Pd catalyst and an appropriate base [1]. Palladium is one of the most effective metals for catalyzing the C-C bonds coupling reactions and has recently gained a great deal of interest [2]. Pd NPs are a viable option in searching for mild reaction conditions and more environmentally friendly procedures because of their great surface-to-volume ratio and incredibly reactive surface atoms compared to the bulk catalyst atoms. The PdII/Pd0 complexes conventionally catalyze coupling reactions in the presence of phosphines or other ligands. Consequently, using Pd NPs enables "ligand-free" synthesis, which reduces costs, simplifies the procedure and facilitates the separation of the resulting product [3-5].
Several homogeneous catalytic systems for the Heck reaction have been reported, but all suffer from poor catalyst separation and recycling, and attempts have been made to successfully resolve the difficulties using heterogeneous catalysts with high activity and minimum leaching [6-8]. Palladium species have been immobilized through a variety of solid supports [9], including magnetic nanoparticles [10], silica gel [11], MOs (metal oxides) [12], MOFs (metal-organic frameworks) [13], CNTs (carbon nanotubes) [14], zeolite [15], alumina [16], graphene [17], and polymers [18]. The use of polymer-bonded NPs takes advantage of the linear polymers, such as excellent thermal and mechanical durability, as well as excellent dispersion provided by nano particles [19, 20]. Similarly, covering Fe3O4 NPs with silica shells and producing magnetic core-shell nanocomposites can prevent aggregation of Fe3O4 NPs and enhance their dispersion, as well as affect surface property and coupling with functional groups arising from polymer components [21-24].
Many studies have reported the high performance of palladium-iron oxide (Pd-Fe3O4) systems in the Heck coupling reaction due to its excellent catalytic properties of Pd and magnetically recoverable feature of Fe3O4. Fe3O4/SiO2-Pd magnetic nanocatalyst with multibranched polyglycerol was created by Zhou et al. and demonstrated high catalytic activity in Heck reaction [25]. Bian and et al. synthesized a hybrid magnetic inorganic/responsive organic polymer nanocomposite via free radical polymerization of sodium acrylate (SAA), 4-vinyl pyridine (4-VP), and undecylenic acid (UDA) onto Fe3O4. The magnetic nanocomposite Fe3O4@P(UDA-co-4-VP-co-SAA) (Fe3O4@PUVS) decorated by Pd to fabricate the Fe3O4@PUVS-Pd nano catalyst, and revealed outstanding catalytic activity in the Suzuki and Heck C-C cross-coupling reactions in aqueous media [26]. Ramazani et al. prepared a robust magnetic palladium catalyst based on artificial metalloenzyme. The catalyst is synthesized through anchoring Pd(2,2′-dipyridylamine)Cl2 supported monomethyl ether poly(ethylene glycol) (mPEG) onto amino modified silica-coated Fe3O4 NPs and demonstrated impressive catalytic activity in Suzuki C-C coupling reaction [27].
As part of our ongoing investigation on Pd catalysts integrated into polymer-grafted silica [28-31], the present work is focused on synthesizing and characterizing a magnetic well defined amino modified thermo-responsive poly (N-isopropylacrylamide) (PNIPAM).
Afterwards, Pd(OAc)2 complexation of this ligand after adding NaBH4 as a reducing agent was performed to achieve the supported catalyst for manufacturing diversely substituted alkenes in Mizoroki-Heck cross-coupling transformation with good to excellent yield and high turnover number (TON).
Experimental
Materials
Azobisisobutyronitrile (AIBN, Merck, 98%) was refined using methanol through recrystallization. N-Isopropylacrylamide (NIPAM) (Acros, 99%) was recrystallized utilizing n-hexane. Citric acid (Sigma-Aldrich 99.5%), vinyltrimethoxysilane (VTES, Sigma-Aldrich, 98%), hydrazine hydrate (Merck, 80% solution in water), and ethylenediamine (Merck, 99%) were used without further purification. 1,4-Dioxane (Sigma-Aldrich, ≥99%) was dried initially utilizing a molecular sieve as preliminary drying agent. The ultimate drying has been performed nearly completely through the distillation of a refluxed solution in the presence of 1% w/v sodium wire prepared with sodium press and 0.2% w/v benzophenone till the benzophenone ketyl radical anion's blue color emerges under an inert environment. Palladium (II) acetate (Pd (OAc)2, 98%, 47% Pd) was purchased from Sigma Aldrich.
Characterization techniques
FT-IR 8300 spectrophotometer-Shimadzu was used to record FT-IR data. To prepare samples, KBr was used for compacting them into a pellet. Bruker Avance DPX instrument (250 MHz in CDCl3) was employed to obtain NMR spectra of the coupling products. To achieve the XRD patterns of MNP and their modified forms, Bruker AXS D8-Advance X-ray diffractometer (λ = 1.541874 Å for Cu Kα radiation and 2θ = 10-90°) was used. An Elemental (CHN) analyzer, LCHN-A10, was applied to measure the amount of carbon, hydrogen, and nitrogen in each sample. The photoluminescence spectrum was derived from a FluoroMax-4 fluorescence spectrometer at an excitation wavelength of 350 nm. Thin-layer chromatography was carried out using silica-gel plates (polygram SIL-UV 254). Scanning electron micrographs were obtained with VEGA3 TESCAN SEM at 20 kV. Gold was sputter-coated onto the specimens to increase the conductivity by KYKY SBC12 sputter coater by applying physical vapor deposition (PVD) technique before SEM analysis. Polymer hydrodynamic sizes were determined using SZ-100 dynamic light scattering (DLS) from aqueous samples with concentration of 1 mg/mL in 2 mL microtubes. Zeta seizer (ZETA-check Particle Charge Reader, Horiba Jobin Jyovin) was applied to estimate the surface charge of the polymer. The amount of grafted PNIPAM onto Fe3O4 MNPs was determined by Perkin Elmer Pyris Diamond thermogravimetric analyzer (TGA). Before TGA analysis, the samples were dried under a vacuum overnight and next scanned at a heating rate of 5 C min-1 under N2 inert gas flow between 25 C to 700 C. Platinum pans were used to analyze samples weighing between 1.5 to 3.5 mg. The saturation magnetizations of bare and surface-functionalized Fe3O4 MNPs were explored by a vibrating sample magnetometer (magnetic daghigh kavir VSM) and samples weighing around 200 mg were used. The Pd amount determination was performed by an inductively coupled plasma implement (Varian, Vista-Pro ICP-OES).
Preparation of super paramagnetic Fe3O4 (I)
In distilled water (40 mL), a combination of FeCl2·4H2O (4.73 mmol, 0.93 g) and FeCl3·6H2O (9.46 mmol, 2.5 g) salts were dissolved. After 15 minutes of ultrasonication, the mixture was transferred to a 250 mL three-necks, round bottom flask equipped with a separatory funnel containing NaOH (40 mL, 1 M) under an Ar environment. During the gradual co-precipitation process, NaOH was added dropwise into the mixture throughout 90 minutes to generate Fe3O4 nuclei. To grow Fe3O4 particles with a spherical form and regulate particle size, the pH of the reaction solution was maintained between 10 to 12. The as-prepared, black product was separated using a small magnet bar, washed multiple times in distilled H2O and EtOH, and next vacuum-dried at 50 C for 9 hours.
Preparation of CQD suspension
As reported previously, amino-rich fluorescent CQD was created by pyrolyzing citric acid and ethylenediamine with the aid of a microwave. Citric acid (1 g, 5.20 mmol) was dissolved in 20 mL water and next, ethylenediamine (0.4679 g, 7.785 mmol) was introduced to produce a transparent and clear solution while stirring vigorously. The solution was then located for 4 minutes in a 700 W microwave oven. The resultant black-brown CQD aqueous solution was cooled to room temperature.
Preparation of CQD coated Fe3O4 MNP (Fe3O4@CQD) (II)
The as-prepared Fe3O4 nanoparticle (0.25 g) was sonicated in water (3 mL) for 10 minutes and next added to the flask with 3.5 mL of the CQD suspension. The suspension was continuously stirred at 60 ᵒC for 1 day. Afterwards, the synthesized Fe3O4@CQD (II) was separated magnetically, and the product was washed multiple times in distilled H2O and EtOH before being vacuum-dried.
Preparation of VTES coated Fe3O4@CQD (Fe3O4@CQD@Si) (III)
Silica-modified magnetite nanoparticle with polymerizable double bonds was produced by treating VTES with the hydroxyl groups on the surface of Fe3O4@CQD. A homogenous suspension was obtained by sonicating 1.0 g of Fe3O4@CQD NPs in 60 mL of EtOH for approximately one hour and then 12 mL of NH3.H2O was added. VTES (6 mL) was introduced to the suspension while constantly stirring for 1 day at 50 ᵒC. The product (Fe3O4@CQD@Si) was then separated using a magnet bar and extensively washed with distilled H2O and EtOH until neutralized and finally vacuum-dried at ambient temperature overnight.
Preparation of thermo-responsive PNIPAM brushes grafted onto modified Fe3O4 MNP (Fe3O4@CQD@Si@PNIPAM) (IV)
In a polymerization tube containing stirrer, azobisisobutyronitrile (AIBN) (0.017 mmol, 2.90 mg) as initiator, N-isopropylacrylamide (8.83 mmol, 1.00 g) as monomer (molar ratio of monomer: AIBN = 150: 0.3) and Fe3O4@CQD@Si (386 mg) were introduced, and then 1.5 mL dry 1,4-dioxane was added. Thereafter, the tube was immersed in liquid nitrogen until the contents were frozen, and then vacuumed for a few minutes to degas. After sealing the polymerization tube, it was heated in oil bath at 85 °C with vigorous stirring for 1 day. Following magnetic separation using a permanent magnet, the resulting product (Fe3O4@CQD@Si@PNIPAM) was completely washed with 1,4-dioxane and vacuum-dried for 24 h at 50 C.
Preparation of Fe3O4@CQD@Si@PNIPAM-NH2 (V)
Compound (IV) (1.38 g) was added gradually to an excess amount of hydrazine monohydrate. (7 ml) in 5 mL 1, 4-dioxane. The suspension was heated for 9 h at 90 °C while thoroughly stirring. A permanent magnet was used for the magnetic separation to obtain the desired amino-modified compound (Fe3O4@CQD@Si@PNIPAM-NH2). The product was washed thoroughly with 1,4-dioxane and then vacuum dried at 60 C.
Preparation of Pd catalyst (Fe3O4@CQD@Si@PNIPAM-NH2/Pd) (VI)
In a reaction flask, the compound (V) (0.22 g) was dispersed in EtOH (4 mL) and then Pd(OAc)2 (0.224 g, 1 mmol) was introduced. The suspension was stirred at ambient temperature for 12 h. The corresponding mixture was next gathered by a permanent magnet and next dried under vacuum overnight. Pd0 NPs supported on Fe3O4@CQD@Si@PNIPAM-NH2 were yielded by adding NaBH4 (10 mg, 0.264 mmol) as a reductant. The magnetic heterogeneous catalytic system was obtained through the complexation of the amino-modified support with Pd. Magnetic separation with a permanent magnet yielded the desired product, which was then vacuum-dried. To measure the Pd loading, the compound (VI) was digested successively in distilled water, HCl and HNO3 (2:1:1) and subjected to ICP. Pd loading was found to be 1.1 mmol of Pd per gram of heterogenous catalyst.
General approach for the Mizoraki-Heck coupling using Pd catalyst (VI)
In a round-bottom flask, a haloarene (1 mmol), K2CO3 (2 mmol), and Pd catalyst (VI) (5 mg, 0.55 mol%) were mixed with DMF (2 mL), and then styrene or n-butylacrylate (1.5 mmol) was introduced to the previous suspension . The suspension was heated at 95 ᵒC with constant stirring for a required time. TLC was used to monitor the reaction. Once the coupling reaction was completed, the suspension cooled to ambient temperature. The catalyst (VI) was simply separated magnetically and the remaining solution was extracted with EtOAc several times. The organic phases were combined, and then evaporated under reduced pressure after drying over anhydrous MgSO4. Purification of the coupling products was then performed using plate chromatography on silica gel with petroleum ether (90): ethyl acetate (10) as the eluent and giving the desired products in 85 to 95% isolated yield. To characterize the coupling products, their spectroscopic data (FT-IR and 1H-NMR) were compared with authentic samples.
Results and Discussion
Preparation and characterization of Fe3O4@CQD@Si@PNIPAM-NH2/Pd catalyst
A thermo-responsive palladium catalyst supported on PNIPAM grafted Fe3O4@CQD@Si was prepared as depicted schematically in Scheme 1. Fe3O4 superparamagnetic nanoparticle (I) was created using salts of Fe2+ and Fe3+ by the co-precipitation procedure. Prepared Fe3O4 nanoparticles were coated with synthesized CQD as the fluorescent agent. CQDs are safe and environmentally friendly for use in medicine and biology, because they release intense fluorescence like QDs and are nontoxic and cheap.
Low toxicity, huge specific surface area, adjustable bandgap, excellent solubility, intense and consistent photoluminescence, and good biocompatibility give CQDs higher economic value than the other QDs, and as a result, they can be utilized as environmentally friendly and sustainable support for coating MNPs [32].
Next, the CQD-modified Fe3O4 NP reacted with vinyltriethylsilane (VTES) to form compound (III) (Fe3O4@CQD@Si) to modify the surface of MNPs by adding polymerizable double bond groups.

Scheme 1. Schematic illustration for the synthesis of Fe3O4@CQD@Si@PNIPAM-NH2/Pd catalyst
Thereafter, PNIPAM bonded Fe3O4 magnetic nanoparticles (Fe3O4@CQD@Si@PNIPAM) (IV) were synthesized using AIBN as initiator and NIPAM as monomer in dry and degassed 1,4-dioxane, through conventional free radical polymerization technique. Finally, the PNIPAM grafted Fe3O4@CQD@Si was modified with hydrazine to transform it to amino-functionalized compound. Eventually, this compound (V) was complexed with Pd(OAc)2 and after the NaBH4 addition as a reducing agent, produce compound (VI) (Fe3O4@CQD@Si@PNIPAM-NH2/Pd NP) in the form of a magnetic heterogeneous catalyst. The optical property of the CQD was evaluated by fluorescence spectroscopy, which revealed two prominent peaks, one at approximately 226 nm owing to the π to π* transition of carbon-carbon and the other at approximately 350 nm due to the n to π* transitions of C=N and C-O. CQDs were excited most efficiently and showed strong blue fluorescence at 360 nm (Figure 1).
FT IR spectra of all synthesized compounds (Fe3O4 (I), Fe3O4@CQD (II), Fe3O4@CQD@Si (III), Fe3O4@CQD@Si@PNIPAM (IV), and Fe3O4@CQD@Si@PNIPAM-NH2/Pd (VI)) are gathered and confirmed their structures (Figure 2). FT IR spectrum of Fe3O4 NPs revealed the absorption peak at 588 cm-1 assign to the stretching vibration of Fe–O bonds and, the broad peak at about 3441 cm−1 is related to O–H stretching of bare Fe3O4 NPs (I) (Figure 2a). The FT-IR spectrum of the Fe3O4@CQD (II) showed the typical absorption of the -NH2 at 1407 cm-1 and 1180 cm-1, and -OH hydroxyl group at 3437 cm-1 (Figure 2b). In the FT-IR spectrum of Fe3O4@CQD@Si (III), frequencies of Si–O–Si and Si–O stretching vibrations emerged at 1134 and 1051cm−1, respectively, which validate the reaction between hydroxyl groups on the Fe3O4 surface with VTES (Figure 2c). The FT IR spectrum for Fe3O4@CQD@Si@PNIPAM (IV) shows expected bands at 3435 cm−1 (N–H), 1634 cm−1 (C=O), 2975 and 1386 cm−1 (C–H bonding) and matching well to the expected structure of the polymer (Figure 2d). The FT-IR spectrum of compound (VI) exhibited the typical absorption of C=O group at 1644 cm-1, -OH hydroxyl group at 3435 cm-1, and aliphatic group C-H at 2964 cm-1 and 2923 cm-1 (Figure 2e).

Figure 1. Fluorescence spectrum of Fe3O4@CQD

Figure 2. (a) FT-IR spectra of (a) Fe3O4 MNPs (I), (b) Fe3O4@CQD (II), (c) Fe3O4@CQD@Si (III), (d) Fe3O4@CQD@Si@PNIPAM (IV), and (e) Fe3O4@CQD@Si@PNIPAM@hydrazine/Pd (VI)
By comparison of magnetic properties of compounds (I) and (IV) by vibration sample magnetometry (VSM), the saturation of the nanoparticles decreased from 150 emu/g for untreated Fe3O4 (I) to 25 emu/g for the Fe3O4@CQD@Si@PNIPAM nanoparticle (IV) (Figure 3). This drop in saturation might result from the surface coating and functionalization of Fe3O4 nanoparticles with polymer chains.
By checking the thermogravimetric analysis (TGA) of compounds (I) and (IV), we can see that the uncoated nanoparticles were thermally stable. Approximately, 85% mass loss of compound (IV) is occurred at two main pyrolysis stages during thermal degradation of organic segments. This compound exhibited good thermal stability under 500 C, and all organic materials were destroyed completely at about 750 °C (Figure 4).

Figure 3. VSM curves for the prepared (A) bare Fe3O4 (I) and (B) Fe3O4@CQD@Si@PNIPAM (IV)

Figure 4. TGA curves for the prepared bare (A) Fe3O4 (I) and (B) Fe3O4@CQD@Si@PNIPAM (IV)
The LCST of thermoresponsive compounds (IV, V, and VI) was measured by heating the samples and record the temopreture at which they begin to shrink. Compounds IV, V, and VI have LCST of approximately 33 C, 36 C, and 40 C, respectively. The increase in LCST from compound (IV) to compound (V) is due to the increase in hydrophilicity of layers. One of the reasons for choosing PNIPAM in the catalyst' structure is due to the fact that hydrophobic portion of PNIPAM begins to shrink after the LCST and the corresponding catalytic reaction in this portion is carried out under mild conditions.

Figure 5. SEM images of (a) Fe3O4 MNPs (I), (b) Fe3O4@CQD (II), (c) Fe3O4@CQD@Si (III), (d) Fe3O4@CQD@Si@PNIPAM (IV), (e) Fe3O4@CQD@Si@PNIPAM-NH2 (VI), and (f) Fe3O4@CQD@Si@PNIPAM-NH2/Pd (V)
The results of CHNS analysis of compounds at various steps of catalyst synthesis were measured, and the increase in the proportion of nitrogen, hydrogen, and carbon content shows the growth of polymer chains and the formation of organic components onto the MNPs.
SEM images were utilized to assessed the morphology and size of the uncoated and coated MNPs' surfaces. The majority of the uncoated nanoparticles are quasi-spherical and have mean sizes of about 0.05 m, as demonstrated in Figure 5. Aggregation was decreased, and nanoparticle dispersion was enhanced after polymer coating. The absence of particle aggregation is likely due to the VTES and PNIPAM coatings, which reduce magnetic interactions between particles (Figure 5).
DLS test was applied to measure the hydrodynamic diameter of bare Fe3O4 and their modified forms (Figure 6). The size distribution graphs for the naked Fe3O4 (I), Fe3O4@CQD (II), Fe3O4@CQD@Si (III), Fe3O4@CQD@Si@PNIPAM (IV), and Fe3O4@CQD@Si@PNIPAM-NH2/Pd (VI) obtained by DLS method according to size distribution data analysis. The Fe3O4 nanoparticles had an average size of 134 nm and relatively low size dispersion (Figure 6a). The average size of Fe3O4@CQD is 178 nm (Figure 6b), Fe3O4@CQD@Si is 674 nm (Figure 6c), Fe3O4@CQD@Si@PNIPAM is 1160 nm (Figure 6d), and Fe3O4@CQD@Si@PNIPAM-NH2/Pd is 1304 nm (Figure 6e). This indicates that the binding of PNIPAM chains onto Fe3O4 nanoparticles and bonding of palladium to polymer chains increase their size.
Dispersion of the nanoparticles and their respective colloidal stability is associated with the surface electric charge of the NPs. Zeta potential measurements revealed value of -33.2 mV for Fe3O4 (I), -39.8 mV for Fe3O4@CQD (II), -49.7 mV for Fe3O4@CQD@Si (III), -54.1 mV for Fe3O4@CQD@Si@PNIPAM (IV) and, -57.4 mV for Fe3O4@CQD@Si@PNIPAM-NH2/ Pd (VI) in water dispersion (see supporting information). Fe3O4 NPs have smaller negative charge on the surface compare to coated Fe3O4 which demonstrate fewer stability and higher tendency to agglomerate over time and form a core structure.
In contrast, the Fe3O4@CQD@Si@PNIPAM nanoparticles have more stability and fewer tendency for agglomeration because of the repulsive force between the particles, which is expressed by a more negative surface charge of the particles. Therefore, it is believed that larger negative surface charge has enhanced the colloidal stability of nanoparticles. The XRD patterns of the prepared Fe3O4 MNPs (I), Fe3O4@CQD (II), Fe3O4@CQD@Si (III), and Fe3O4@CQD@Si@PNIPAM (IV) confirm the formation of inverse spinel crystal structure of magnetite Fe3O4. The position and also respective intensity of all detected diffraction peaks are the same as with JCPDS Card No. 75-0033 properly (Figure 7).
The diffraction peaks corresponding to 2Ө=30.3°, 35.65°, 43.35°, 53.4°, 57.5 and 62.95° manifested by their indices (210), (311), (400), (422), (511), and (440), respectively, are characteristic peaks of the Fe3O4 nanoparticles. The quite identical diffraction peaks of compounds (II, III, and IV) with Fe3O4 (I) suggest that the modification procedure had not affected the positions of the peaks and also particles crystal structure. Using the Scherrer equation, the crystallite size of Fe3O4 nanoparticles (I) (Figure 7a), Fe3O4@CQD (II) (Figure 7b), Fe3O4@CQD@Si (III) (Figure 7c) and, Fe3O4@CQD@Si@PNIPAM (IV) (Figure 7d) were found to be about 138, 193, 308 and, 430 nm, respectively.
.jpg)
.jpg)
Figure 6. DLS of (a) Fe3O4 MNPs (I), (b) Fe3O4@CQD (II), (c) Fe3O4@CQD@Si (III), (d) Fe3O4@CQD@Si@PNIPAM (IV), and (e) Fe3O4@CQD@Si@PNIPAM@hydrazine/Pd (VI)

Figure 7. XRD patterns of (a) Fe3O4 (I), (b) Fe3O4@CQD (II), (c) Fe3O4@CQD@Si (III), (d) Fe3O4@CQD@Si@PNIPAM (IV), and (e) Fe3O4@CQD@Si@PNIPAM-NH2/Pd (VI)
Application of Fe3O4@CQD@Si@PNIPAMNH2/Pd catalyst in Heck coupling reaction
To check the potency of the designed Pd catalyst (VI), it was explored in the Heck cross-coupling reaction of various haloarenes with n-butyl acrylate or styrene. To optimize the reaction conditions, the reaction between iodobenzene with n-butyl acrylate was firstly investigated as a model reaction for catalyst (VI). The results are presented in Table 1. Initially, we investigate the impact of various bases (K2CO3, NEt3, Na2CO3, KOH, and NaOH) and solvents (DMF, H2O, EtOH, toluene, DMF/H2O, and EtOH/H2O), various temperature from 55 to 120 C and varying amount of Pd catalyst (2-20 mg) to determine how the divers condition affected the outcomes. Rising the temperature from 55 °C to 95 °C improved the yield of the product, but there was not any significant improvement by changing the temperature from 95 °C to 120 °C (Table 1, entry 1-3). Among the solvents including EtOH, EtOH/H2O (1:1), toluene, DMF, DMF/H2O (1:1), and H2O, DMF yield the most percent of the product in 1 hour (entry 5 and entries 7-11). Base free condition (entry 16) indicates that Heck reaction needs a base for reaction progress. Among various bases tested, K2CO3 provided a higher yield (entry 5). Other bases like Na2CO3, KOH, NaOH, and NEt3 were explored and proved lower efficiency (entries 12-15). In addition, we investigated the influence of Pd catalyst amount on the coupling reaction between phenyl iodide and n-butyl acrylate (entries 2 and 4-6). The catalyst loading can be decreased to 2 mg Pd by extending the time of the reaction to 120 min and 80% conversion of iodobenzene was obtained. Eventually, the optimal conditions for this Pd catalyst were determined to be 5 mg of catalyst in DMF and K2CO3 as a base at 95 C.
Table 1. Optimization of base, solvent, and amount of catalyst for the reaction of iodobenzene with n-butyl acrylate in the presence of Pd catalyst (VI)a
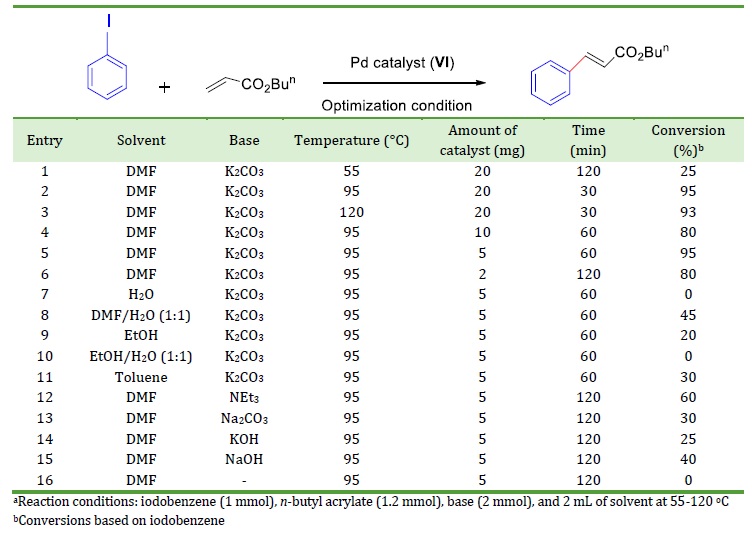
Table 2. Heck reaction of n-butyl acrylate or styrene with different aryl halides in the presence of Pd catalyst (VI)

To probe the efficiency of the developed Pd catalyst, the Mizoroki-Heck cross-coupling reaction of several haloarenes with n-butyl acrylate (n-Bu acrylate) or styrene (St) was conducted and a range of substituted alkenes yielded. The reaction was carried out in DMF in the presence of K2CO3 and Pd catalyst at 95 oC. Applying Pd catalyst, coupling reactions between various aryl halides and n-Bu acrylate or St demonstrated the universality of this reaction system (Table 2). Aryl iodides with electron-deficient and electron-rich substitutes react well with n-butyl acrylate and styrene to create products with high-quality outcomes. Electron-withdrawing groups shorten the time of the reaction, whereas electron-donating groups lengthen it. Thus, for example, 1-iodo-4-nitrobenzene react more quickly than 4-iodotoluene in the reaction with styrene (Table 2, entries 9 and 11).
Conclusion
The present study reported the synthesis and characterization of Pd catalyst based on the modified thermo-responsive PNIPAM grafted Fe3O4@CQD@Si via conventional free radical polymerization technique. Initially, Fe3O4 NPs were created by the co-precipitation method, and then CQD, as green and sustainable support, was used to coat Fe3O4 NPs instead of the conventional SiO2. Chemical modification of Fe3O4@CQD with polymerizable groups was performed through the reaction with VTES. Finally, PNIPAM was grafted to the modified Fe3O4, and then it was modified with hydrazine to convert it to an amino ligand. The amino support was subsequently coupled with Pd through reaction with Pd(OAc)2, and then adding NaBH4 as a reducing agent to obtain the heterogeneous catalytic system. All materials are characterized through a wide range of methods, such as FT-IR, XRD, CHN, SEM, DLS, zeta potential, VSM, and TGA analysis techniques. This catalytic system was optimized for producing a variety of substituted alkenes in Mizoroki-Heck coupling reaction in good to outstanding yield. Easy purification of the substituted alkenes due to the convenient recyclability of the catalyst is the main characteristic of this process.
Acknowledgements
The authors would greatly have appreciated the partial support of this study by Shiraz University Research Council.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Orcid
Soheila Ghasemi
https://orcid.org/0000-0003-4469-0333
How to cite this manuscript: Soheila Ghasemi*, Fatemeh Badri, Hadieh Rahbar Kafshboran. Pd Catalyst Supported Thermo-Responsive Modified Poly(N-isopropylacrylamide) Grafted Fe3O4@CQD@Si in Heck Coupling Reaction. Asian Journal of Green Chemistry, 8(1) 2024, 39-56. DOI: 10.22034/ajgc.2023.408188.1401