Document Type : Original Research Article
Authors
Department of chemistry, College of science, University of Baghdad, Baghdad, Iraq
Abstract
A fast and sensitive method for detecting resorcinol by producing yellowish-green color species using the reaction of potassium dichromate with resorcinol in a sulfuric acid medium. NAG-4SX3-3D analyzer was used to evaluate the transducer energy response. To improve the sensitivity of the newly developed approach, the relevant parameter was explored. The linear range (0.05-32) mmol.L-1 for resorcinol measurement and RSD percent for the repetition (n=6) was significantly lower than 0.3% for (0.7, 23 millimol.L-1) with L.O.D.=48.1687 ng/sample from the progressive dilution across the calibration graph's lowest concentration linear dynamic range (r=0.9997), (correlation coefficient), percentage linearity (R2 %=99.95). The proposed approach was compared to the previous technique (UV-spectrophotometric at λmax=273 nm). It can be concluded that, in addition to the technique's sensitivity (developed) and the use of a few chemicals, it outperforms the 10 mm irradiation of the classic reference method. Furthermore, continual dilution in CFIA enables the handling of high or low concentrations, opening up a larger range of applications. Based on the foregoing, the developed methodology is judged to be the most suited for resorcinol molecules compared to the reference techniques.
Graphical Abstract
Keywords
Introduction
Resorcinol (Res) 1,3-isomer of benzenediols, as illustrated in Scheme 1, is a dihydric phenol exhibiting characteristic phenol reactivity. It is a monoaromatic compound having two hydroxyl groups in meta position to each other that may be found in fossil fuels in the heartwood of Artocarpus and Moraceae species. Resorcinol ring-containing components have been discovered in various natural compounds, including plant phenolics which include resorcinol ring-containing components and are widely distributed in nature [1]. Resorcinol has widely utilized in various industries, including the production of rubber products [2]. Resorcinol is used in the rubber industry as a primer in latex fabric coating [3] and as a glue in rubber product manufacture, such as when attaching the ends of a conveyor belt. It is included in many hair color products because of its unique functioning as a coupler. It is also found in medicinal items [4] such as eye drops that can be purchased without a prescription and a variety of ointments for disorders as psoriasis and acne.
Local hyperemia, corrosion, itching, dermatitis, and the loss of superficial layers of the skin may occur when solutions or salves containing 3 to 25% resorcinol are used [5]. It causes skin and eye discomfort if eaten, absorbed, or breathed through the skin. The drug may have effects on the blood, inducing methaemoglobin production. It is considered goitrogenic, causing hypothyroidism. Resorcinol was discovered in an aqueous solution [6] as an intermediary in the anaerobic decomposition of methoxyphenol and as an irradiation product of 3-chlorophenol.
There are many methods for the determination of resorcinol, such as electromagnetic sensor [7], electrochemical activation of graphene sheets embedded in carbon films [8], and a surfactant enhanced graphene paste electrode [9], electrochemical oxidation [10], electrochemical detection [11].
.jpg)
Scheme 1. Structure of resorcinol
Experimental
Materials and methods
The solutions were produced using distilled water, and all of the compounds used were analytical reagent grade. By dissolving 11 g in 500 mL of distilled water, a 0.2 M standard solution of Resorcinol C6H4 (OH)2 with molecular weight 110.1 g.mol-1, BDH was made. A standard solution of potassium dichromate K2Cr2O7 with a molecular weight of 294.22 g.mol-1 Hopkin and Williams LTD was made by dissolving 18.38 g of potassium dichromate in 250 mL of distilled water.
Apparatus
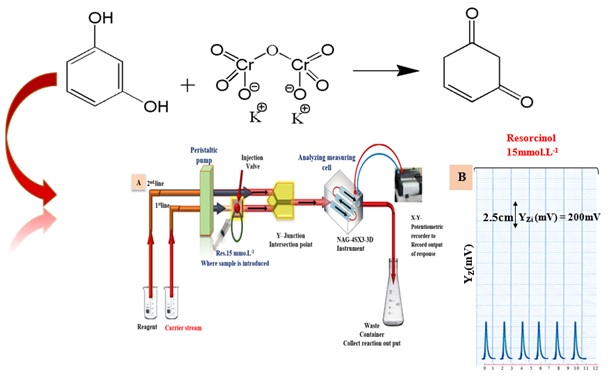
A flow cell manufactured from a handmade NAG-4SX3-3D analyzer [12] was used to obtain the output from the attenuation of incident light (0–180 ͦ). Figure 1a Potentiometric recorders were used to record the output signals (Siemens, Germany). A six-port injection valve and an ismatic peristaltic pump with a sample loop (Teflon, variable length), the traditional procedures were carried out using a Uv-spectrophotometric (Shimadzu, Japan).
Methodology
A manifold design for determining an aromatic organic chemical (resorcinol) Figure 1a by forming colored species with potassium dichromate. It consists of a two-line manifold system attached to the NAG-4SX3-3D analyzer. A sample segment introduction unit (injection valve with load injection location) is included in the system, allowing a certain quantity to be injected repeatedly with flawless dependability. The first line provides distilled water as a carrier stream for the sample zones of resorcinol 15 mmol.L-1 with 115 µL as a sample volume to meet with potassium dichromate in the second line at a 2.8 mL.min-1 flow rate for each line by Y-junction point before being introduced to the NAG-4SX3-3D analyzer. The x-t potentiometric record output was used to quantify the transducer energy response for the attenuation of incident light on colored species for a yellowish-green species which formed. Each solution was investigated three times. Scheme 2 proposed a mechanism for the oxidation of resorcinol by potassium dichromate. Figure 2 displays the output Yz (mV) vs. tmin (dmm) of the NAG-4SX3-3D analyzer transducer for 15 mmol.L-1 of resorcinol pharmaceuticals over time. The anther considers the synchronization of system outputs a new method in the NAG-4SX3-3D analyzer.
.jpg)
Scheme 2. Proposed reaction for resorcinol-K2Cr2O7 system
Result and Discussion
The flow injection manifold system (Figure 1a) was used to investigate chemical and physical factors to determine the circumstances that would yield the reaction product, yellowish-green species, with the highest repeatability and sensitivity. The most straightforward strategy to optimize these variables was to keep them all constant while modifying them one by one.
Chemical variables
Effect of variable concentration of potassium dichromate
The measurements were carried out under the following conditions: A series of potassium dichromate solutions were generated by diluting the stock solution with distilled water to obtain concentrations ranging from 10 to 150 mmol.L-1. Each line was measured three times with a reagent concentration of 15 mmol.L-1, a sample volume of 115 µL, an open valve mode, and a flow rate of 2.8 mL.min-1 for the carrier stream (distilled water) and reagent. Figure 1a reveals the response profile for this investigation which shows that the energy transducer response fluctuates with potassium dichromate concentration when using the NAG-4SX3-3D analyzer.
It was noticed that an increase in the response of the colored species with an increase of potassium dichromate concentration up 100 mmol.L-1, more than 100 mmol.L-1 led to broadening in high peak maxima and increase the peak base width (ΔtB), this due to increase the intensity of colored species which play as an internal filter in front of detector that prevents remaining light after absorption process from passes to solar cell. Therefore, 100 mmol.L-1 of potassium dichromate was selected as an optimum concentration in the following studies to give a maximum peak height and sharpness. The obtained result was tabulated in Table 1a, while Figure 1b and Table 1b demonstrate the segmentation pattern for selecting the optimum segment of Res. Systems, segment S3 (i.e., 70-150 mmol.L-1) for the resorcinol–K2Cr2O7 system.
.jpg)
Figure 1. a) Response profile of potassium dichromate concentrations, b) Output of (S/N) energy transducer response, and three data points as one segment with optimum choice
.jpg)
Figure 2. a) Diagram of manifold used for assessment NAG-4SX3-3D via reaction of resorcinol 15 mmol.L-1 with Potassium dichromate 50 mmol.L-1 to form yellowish green colored species. b) Profile of preliminary repeated experiments for assessment NAG-4SX3-3D instrument via reaction resorcinol with potassium dichromate to form yellowish species
Table 1. a) Effect of potassium dichromate concentration on precipitate formation of resorcinol, b) segmentation pattern slop-intercept with selection optimum segment for res-potassium dichromate system
.jpg)
Effect of various medium
Using certain resorcinol system conditions, resorcinol 15 mmol.L-1-K2Cr2O7, 100 mmol.L-1, and a 115 µL sample volume, 2.8 mL/min-1 was the flow rate. The impact of various solutions was examined using a carrier stream. In addition to the aqueous medium, (CH3COOH, Tartaric acid, Ascorbic acid, HCl, HNO3, H2SO4, KCl, CH3COONH4) with a 0.4 mmol.L-1 concentration. Figure 3 shows that the investigated media generate a reduction in S/N-response. This might be due to a rise in agglomeration or the growth in aggregate density and compactness with one another, which leads to an increase in S/N response due to the effect of tiny solid particulate formation that causes a decrease in inter-spatial distances and increases incident light attenuation, this results in an increase in incident light intensity because there will be more empty spaces in between agglomerates of particulate except H2SO4. In this research study, H2SO4 medium was chosen as the best carrier stream for resorcinol beca